A cost-effective nanotube-based catalyst for producing hydrogen fuel
July 28, 2014
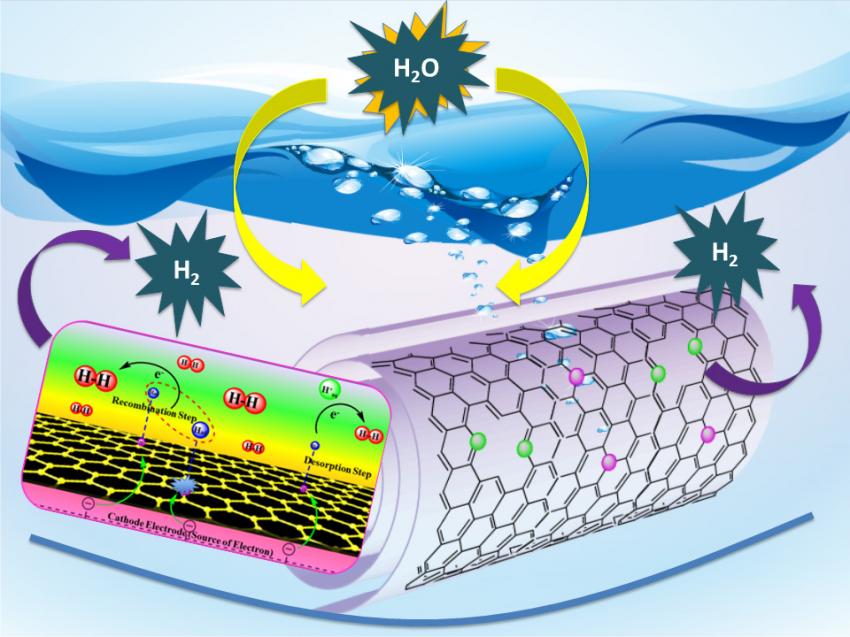
A new technology based on carbon nanotubes promises commercially viable hydrogen production from water (credit: Tewodros Asefa)
Rutgers researchers have used carbon nanotubes as a catalyst for splitting water into hydrogen and oxygen, which could replace expensive platinum for making clean-burning hydrogen fuel — which could one day replace expensive, environmentally harmful fossil fuels.
The Rutgers technology is also far more efficient than other low-cost catalysts investigated to date for electrolysis reactions, which use electric currents to split water molecules into hydrogen and oxygen, the researchers say.
Finding ways to make electrolysis reactions commercially viable is important because processes that make hydrogen today start with methane — itself a fossil fuel. So the need to consume fossil fuel negates current claims that hydrogen is a “green” fuel.
Electrolysis, however, could produce hydrogen using electricity generated by renewable sources, such as solar, wind and hydro energy, or by carbon-neutral sources, such as nuclear energy. And even if fossil fuels were used for electrolysis, the higher efficiency and better emissions controls of large power plants could give hydrogen fuel cells an advantage over less efficient and more polluting gasoline and diesel engines in millions of vehicles and other applications, the researchers claim.
Earth-abundant elements
In a recent scientific paper published in Angewandte Chemie International Edition, Tewodros (Teddy) Asefa, associate professor of chemistry and chemical biology in the School of Arts and Sciences, and his colleagues reported that their cobalt-embedded, nitrogen-rich carbon nanotubes (Co-NRCNTs) — containing only earth-abundant elements (C, N and Co) — to catalyze hydrogen with efficiency close to that of platinum.
The paper also noted that the new material functions well in acidic, neutral, and basic conditions. That’s important because the water-splitting reaction actually combines two different reactions (based on hydrogen and oxygen): the hydrogen reaction requires an acidic condition, while the oxygen reaction requires neutral or basic media. But most of the recently developed catalysts for the hydrogen reaction function well only in acidic media.
The researchers have filed for a patent on the Co-NRCNT catalyst, which is available for licensing or research collaborations through the Rutgers Office of Technology Commercialization. The National Science Foundation funded the research.
Will Toyota sell more than 1,000 hydrogen fuel cell vehicles (FCV) in the US before the end of 2015?
These two questions are taken from SciCast, a research project run by George Mason University to forecast the outcomes of key issues in science and technology. SciCast is based on the idea that the collective wisdom of an informed and diverse group is often more accurate at forecasting the outcome of events than that of one individual expert. Funding is provided by IARPA, the Intelligence Advanced Research Projects Activity.
To make a forecast on SciCast, first, sign up. Click on any of the questions to read more about it and make a forecast. Tutorials are here or send questions to [email protected]. Would you like to see a question of your own on SciCast? Join other SciCast forecasters on Spark, SciCast’s crowdsourced forecast publishing platform. Propose a question.
Abstract of Angewandte Chemie International Edition paper
Despite being technically possible, splitting water to generate hydrogen is still practically unfeasible due mainly to the lack of sustainable and efficient catalysts for the half reactions involved. Herein we report the synthesis of cobalt-embedded nitrogen-rich carbon nanotubes (NRCNTs) that 1) can efficiently electrocatalyze the hydrogen evolution reaction (HER) with activities close to that of Pt and 2) function well under acidic, neutral or basic media alike, allowing them to be coupled with the best available oxygen-evolving catalysts—which also play crucial roles in the overall water-splitting reaction. The materials are synthesized by a simple, easily scalable synthetic route involving thermal treatment of Co2+-embedded graphitic carbon nitride derived from inexpensive starting materials (dicyandiamide and CoCl2). The materials’ efficient catalytic activity is mainly attributed to their nitrogen dopants and concomitant structural defects.