A high-resolution nanoscale window to the live biological world
December 27, 2012
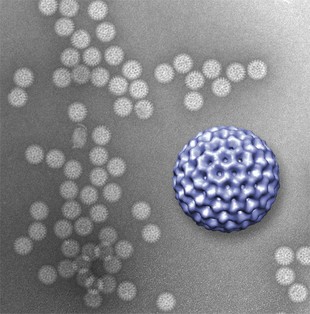
A novel microfluidics platform allowed viewing of structural details of rotavirus double-layered particles; the 3-D graphic of the virus, in purple, was reconstructed from data gathered by the new technique and is ~80 nm. in diameter (credit: Virginia Tech)
Investigators at the Virginia Tech Carilion Research Institute have invented a way to directly image biological structures at nanometer-resolution in their natural habitats (a liquid environment).
The technique is a major advancement toward the ultimate goal of imaging biological processes in action at the atomic level.
The technique uses two silicon-nitride microchips with windows etched in their centers and pressing them together until only a 150-nanometer space between them remains.
The researchers then fill this pocket with a liquid resembling the natural environment of the biological structure to be imaged, creating a microfluidic chamber.
Then, because the movement of free-floating structures yield images with poor resolution, the researchers coat the microchip’s interior surface with a layer of natural biological tethers, such as antibodies, which naturally grab onto a virus and hold it in place.
The ultimate goal is live electron-microscope imaging of molecular mechanisms, such as viral assembly pathways and viral entry into host cells.
“This exciting new frontier would allow investigators to examine biological processes in solution with a remarkable new level of resolution,” the researchers suggest in an open-access paper, “Visualizing viral assemblies in a nanoscale biosphere,” in Lab on a Chip .
Imaging a live rotavirus
The researchers tested the imaging systems with rotavirus double-layered particles.
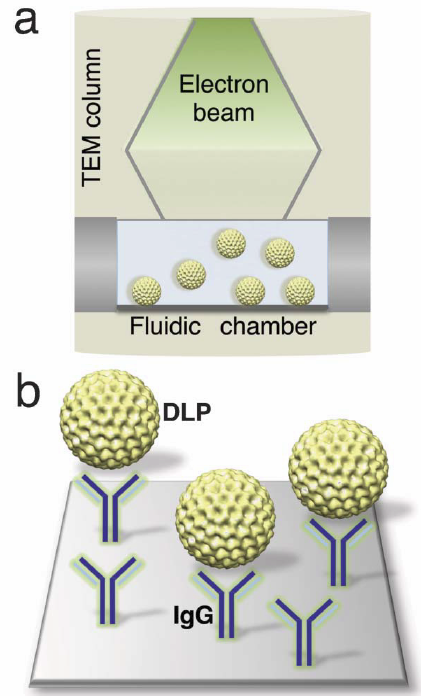
Schematic of a microfluidic chamber (a) that accommodates biological assemblies (yellow) in solution while inside a transmission electron microscope column. (b) Magnified view of the chamber surface decorated with IgG antibodies (green) against the outer capsid protein of the rotavirus double-layered particles (DLP) (credit: Virginia Tech)
Rotavirus is the most common cause of severe diarrhea among infants and children. By the age of five, nearly every child in the world has been infected at least once. And although the disease tends to be easily managed in the developed world, in developing countries rotavirus kills more than 450,000 children a year.
At the second step in the pathogen’s life cycle, rotavirus sheds its outer layer, which allows it to enter a cell, and becomes what is called a double-layered particle. Once its second layer is exposed, the virus is ready to begin using the cell’s own infrastructure to produce more viruses. It was the viral structure at this stage that the researchers imaged in the new study.
The researchers coated the interior window of the microchip with antibodies to the virus. The antibodies, in turn, latched onto the rotaviruses that were injected into the microfluidic chamber and held them in place.
The researchers then used a transmission electron microscope to image the prepared slide.
The experiment gave results that resembled those achieved using traditional freezing methods to prepare rotavirus for electron microscopy, proving that the new technique can deliver accurate results — without killing the specimen first.
The next step is to continue to develop the technique with an eye toward imaging biological structures dynamically in action. Specifically, the researchers are looking to understand how rotavirus assembles, so as to better know and develop tools to combat this particular enemy of children’s health.