A multifunctional nano carrier to detect, diagnose, and deliver drugs to cancer cells
October 31, 2013
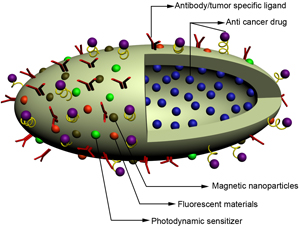
(Credit: University of Cincinnati)
A unique nanostructure developed by a team of international researchers* promises improved all-in-one detection, diagnoses, and drug-delivery treatment of cancer cells.
It can carry a variety of cancer-fighting materials on its double-sided (Janus) surface and within its porous interior and can:
- Transport cancer-specific detection nanoparticles and biomarkers to a site within the body, e.g., the breast or the prostate. This promises earlier diagnosis than is possible with today’s tools.
- Attach fluorescent marker materials to illuminate specific cancer cells, so that they are easier to locate and find for treatment by drug delivery or surgery.
- Deliver anti-cancer drugs for pinpoint-targeted treatment of cancer cells. Currently, a cancer treatment like chemotherapy affects not only cancer cells but healthy cells as well, leading to serious and often debilitating side effects.
- Carry multiple components in a single assembly and function in an intelligent manner, thanks to its functionally and chemically distinct surface.
How it works
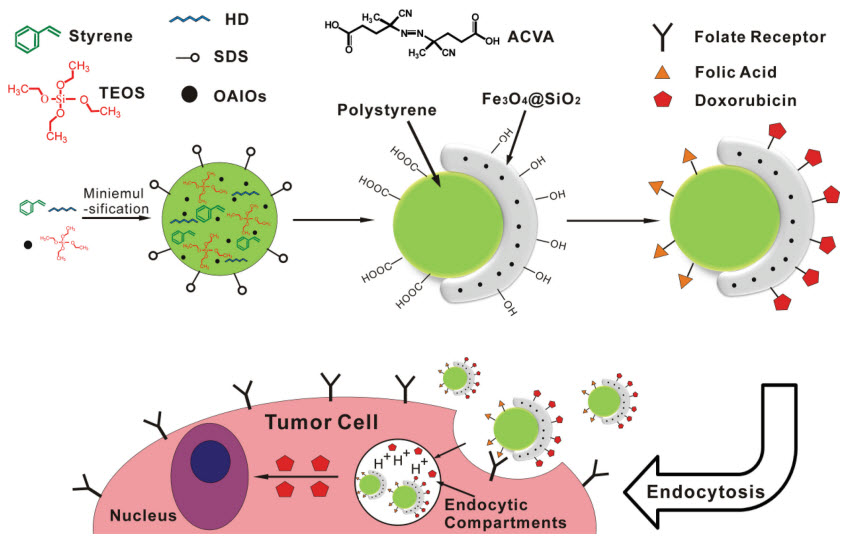
Schematic diagram illustrating synthesis of Polystyrene/Fe3O4@SiO2 superparamagnetic Janus nanocomposites and the proposed mechanisms for tumor-cell targeting and stimulus-induced drug release (credit: Feng Wang et al./Advanced Materials)
“In this effort, we’re using existing basic nano systems, such as carbon nanotubes, graphene, iron oxides, silica, quantum dots and polymeric nano materials to create an all-in-one, multidimensional and stable nano carrier that will provide imaging, cell targeting, drug storage and intelligent, controlled drug release,” said University of Cincinnati professor of materials science and engineering Donglu Shi.
He also said the nano carrier’s promise is currently greatest for cancers that are close to the body’s surface, such as breast and prostate cancer.
Currently, the most common methods used in cancer diagnosis are magnetic resonance imaging (MRI), positron emission tomography (PET), and computed tomography (CT), which are costly and time-consuming to use, the researchers note.
In addition, nanotechnology like the Janus structure would better control the drug dose, since that dose would be targeted to cancer cells, so anticancer drugs could be used much more efficiently. That would, in turn, lower the total amount of drug administered, according to the researchers.
“Currently we’re moving forward to have this nano-carrier tested in-vivo. We’re also trying to scale up the production of the Janus nanocomposites. We already have the prototype of analysis kit using this nanocomposite and we’re planning on filing a patent on it,” researcher Feng Wang, a postdoc at the University of Houston, explained to KurzweilAI.
This research will be presented as an invited talk on Oct. 30, 2013, at the annual Materials Science & Technology Conference in Montreal, Canada.
The work was supported by via grants from the National Science Foundation (No. IOS-0843424); National Natural Science Foundation of China (No. 51003077, No. 51173135, and No. 51073121); Shanghai Nano-program (No. 11nm0506011); and the Fundamental Research Funds for the Central Universities.
* Researchers are Feng Wang, a former UC doctoral student and now a postdoc at the University of Houston; Donglu Shi, professor of materials science and engineering at UC’s College of Engineering and Applied Science (CEAS); Yilong Wang of Tongji University, Shanghai, China; Giovanni Pauletti, UC associate professor of pharmacy; Juntao Wang of Tongji University, China; Jiaming Zhang of Stanford University; and Rodney Ewing of Stanford University.
——————————————————————————————————————————————————————————
Abstract of Advanced Materials paper
Folic acid (FA) and doxorubicin (DOX) are coupled separately onto Fe3O4@SiO2 and polystyrene surfaces of a unique polystyrene/Fe3O4@SiO2 Janus structure. This super-paramagnetic, dual-functionalized Janus nanocomposite enables effective tumor cell targeting and internalization via the folate receptor, and induces significant cancer cell death by controlled, stimulus-induced drug release under acidic conditions in endosomal compartments.