Bioengineers create rewritable digital data storage in DNA
May 22, 2012
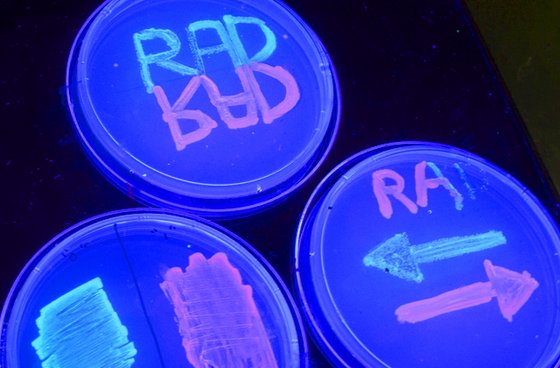
Under ultraviolet light, petri dishes containing cells glow red or green depending upon the orientation of a specific section of genetic code inside the cells' DNA. The section of DNA can be flipped back and forth using the RAD technique. (Credit: Stanford University)
A method for repeatedly encoding, storing and erasing digital data within the DNA of living cells, using natural enzymes adapted from bacteria — the genetic equivalent of a bit — has been developed by Stanford University scientists in the Department of Bioengineering, a joint effort of the School of Engineering and the School of Medicine.
“Programmable data storage within the DNA of living cells would seem an incredibly powerful tool for studying cancer, aging, organismal development and even the natural environment,” said Stanford assistant professor Drew Endy. Researchers could count how many times a cell divides, for instance, and that might someday give scientists the ability to turn off cells before they turn cancerous.
Non-volatile memory
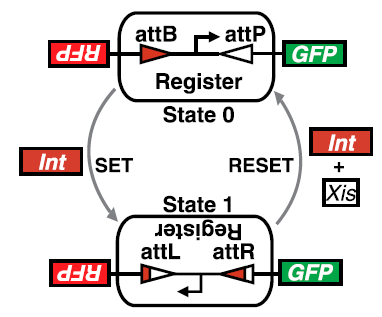
Architecture, mechanisms, and operation of a recombinase addressable data (RAD) module. The DNA inversion RAD module is driven by two generic transcription input signals, set and reset. A set signal drives expression of integrase that inverts a DNA element serving as a genetic data register. Flipping the register converts flanking attB and attP sites to attL and attR sites, respectively. A reset signal drives expression of integrase and excisionase and restores both register orientation and the original flanking attB and attP sites. The register itself encodes a constitutive promoter which initiates strand-specific transcription. Following successful set or reset operations, mutually exclusive transcription outputs “1” or “0” are activated, respectively. For the RAD module developed here, a “1” or “0” register state produces red or green fluorescent protein, respectively. (Credit: Stanford University/PNAS)
In the computer world, their work would form the basis of what is known as non-volatile memory — data storage that can retain information without consuming power. In biotechnology, it is known as “recombinase-mediated DNA inversion,” after the enzymatic processes used to cut, flip and recombine DNA within the cell.
The team calls its device a “recombinase addressable data” module, or RAD for short. They used RAD to modify a particular section of DNA with microbes that determines how the one-celled organisms will fluoresce under ultraviolet light. The microbes glow red or green depending upon the orientation of the section of DNA. Using RAD, the engineers can flip the section back and forth at will.
To make their system work, the team had to control the precise dynamics of two opposing proteins, integrase and excisionase, within the microbes. “Previous work had shown how to flip the genetic sequence irreversibly — in one direction through the expression of a single enzyme,” said Stanford postdoctoral scholar Jerome Bonnet, “but we needed to reliably flip the sequence back and forth, over and over, in order to create a fully reusable binary data register.”
The researchers found it was fairly easy to flip a section of DNA in either direction. “But we discovered time and again that most of our designs failed when the two proteins were used together within the same cell,” said Endy. “Ergo: Three years and 750 tries to get the balance of protein levels right.”
Going from bit to byte
Bonnet has now tested RAD modules in single microbes that have doubled more than 100 times and the switch has held. He has likewise switched the latch and watched a cell double 90 times, and set it back. The latch will even store information when the enzymes are not present. In short, RAD works. It is reliable and it is rewritable.
“One of the coolest places for computing,” Endy said, “is within biological systems.” His goal is to go from the single bit he has now to eight bits — or a “byte” — of programmable genetic data storage.
To get there, however, science will need many new tools for engineering biology, he added, but it will not be easy. “Such systems will likely be 10 to 50 times more complicated than current state-of-the-art genetic engineering projects,” he said. “We’re probably looking at a decade from when we started to get to a full byte.
The research was funded by the National Science Foundation’s Synthetic Biology Engineering Research Center, and by fellowship grants from Stanford’s Center for Longevity and its Bio-X program.
Ref.: Jerome Bonnet, Pakpoom Subsoontorn, and Drew Endy, Rewritable digital data storage in live cells via engineered control of recombination directionality, Proceedings of the National Academy of Sciences, 2012 DOI: 10.1073/pnas.1202344109 (open access)