Brain-machine interface allows precise control of coma and general anesthesia or sedation
November 13, 2013
Researchers have developed a brain-machine interface (BMI) that monitors a patient’s brain activity and adjusts the anesthetic infusion rate to precisely control the level of brain activation in a medically induced coma or for general anesthesia, according to a study published online Oct. 31 in the journal PLoS Computational Biology (open access).
The team includes lead author Maryam Shanechi, a visiting professor and an incoming assistant professor in Cornell’s School of Electrical and Computer Engineering, and Emery Brown and colleagues from Massachusetts General Hospital and Massachusetts Institute of Technology.
“Our work demonstrates the feasibility of automatic, reliable and accurate control of medical coma that can provide considerable therapeutic benefits,” Shanechi told KurzweilAI.
The BMI system would replace monitoring a patient’s brain activity with an electroencephalogram (EEG) and adjusting the anesthetic infusion rate — equivalent to manually flying a plane on a fixed trajectory for several days, rather than using autopilot, and moreover makes accurate control infeasible, the researchers say. It was tested on rodents, but could be used with human patients, the study suggests.
Better brain protection for general anesthesia and sedation too
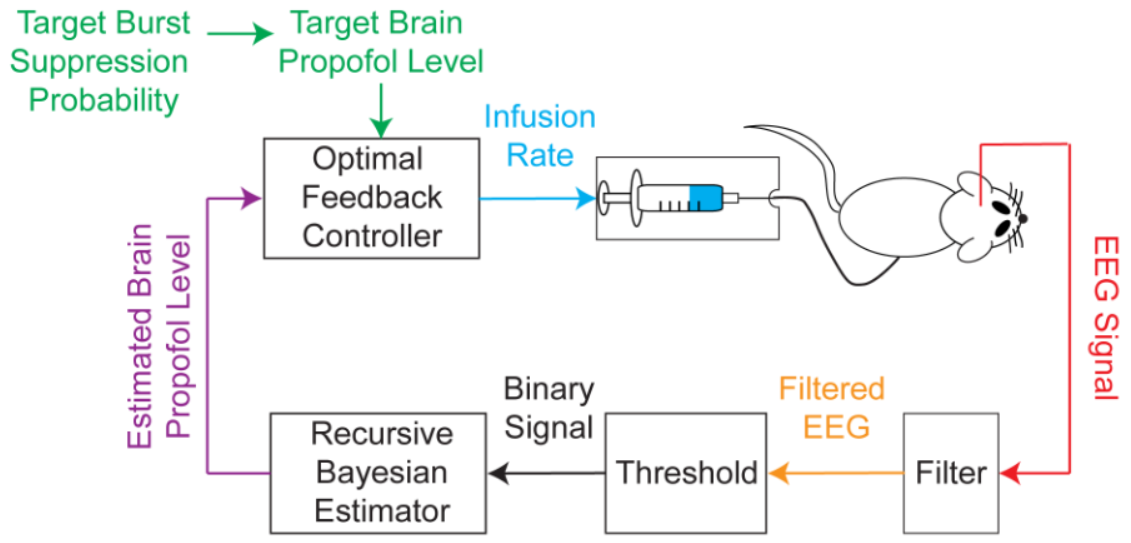
The BMI system records the EEG, segments the EEG into a binary time-series by filtering and thresholding, estimates the BSP or equivalently the effect-site concentration level based on the binary-time series, and then uses this estimate as feedback to control the drug infusion rate (credit: Shanechi MM et al./PLoS Computational Biology)
The BMI’s algorithms use the EEG signal to estimate the level of brain activation in coma, then use this estimate as feedback to control the drug infusion rate in real time.
Reliable and accurate control of medically induced comas could offer the possibility of ensuring adequate brain protection for patients, while using the least amount of anesthetic. It would also make it easier to induce periodic arousals in patients to conduct neurological assessments and prevent anesthetic overdose.
The study further suggests that this system could be adapted to precisely control states of general anesthesia and sedation for patients requiring surgical or nonsurgical procedures. Such an anesthesia delivery system could significantly alter the way anesthesia is administered, particularly during long surgeries.
Automated control may also allow for more efficient use of intensive care unit personnel, as a single nurse per shift would not be solely dedicated to manually managing the anesthetic infusion rate for a single patient for several days, the team suggests.
Abstract of PLoS Computational Biology paper
Medically-induced coma is a drug-induced state of profound brain inactivation and unconsciousness used to treat refractory intracranial hypertension and to manage treatment-resistant epilepsy. The state of coma is achieved by continually monitoring the patient’s brain activity with an electroencephalogram (EEG) and manually titrating the anesthetic infusion rate to maintain a specified level of burst suppression, an EEG marker of profound brain inactivation in which bursts of electrical activity alternate with periods of quiescence or suppression. The medical coma is often required for several days. A more rational approach would be to implement a brain-machine interface (BMI) that monitors the EEG and adjusts the anesthetic infusion rate in real time to maintain the specified target level of burst suppression. We used a stochastic control framework to develop a BMI to control medically-induced coma in a rodent model. The BMI controlled an EEG-guided closed-loop infusion of the anesthetic propofol to maintain precisely specified dynamic target levels of burst suppression. We used as the control signal the burst suppression probability (BSP), the brain’s instantaneous probability of being in the suppressed state. We characterized the EEG response to propofol using a two-dimensional linear compartment model and estimated the model parameters specific to each animal prior to initiating control. We derived a recursive Bayesian binary filter algorithm to compute the BSP from the EEG and controllers using a linear-quadratic-regulator and a model-predictive control strategy. Both controllers used the estimated BSP as feedback. The BMI accurately controlled burst suppression in individual rodents across dynamic target trajectories, and enabled prompt transitions between target levels while avoiding both undershoot and overshoot. The median performance error for the BMI was 3.6%, the median bias was -1.4% and the overall posterior probability of reliable control was 1 (95% Bayesian credibility interval of [0.87, 1.0]). A BMI can maintain reliable and accurate real-time control of medically-induced coma in a rodent model suggesting this strategy could be applied in patient care.