Creating a low-cost, flexible touchscreen
September 5, 2013

Comparing two nanowire touchscreen designs. Left: high transparency but high resistance; right: low resistance but low transparency (credit: University of Pennsylvania)
Future touchscreens* will be flexible, cheap, and give you finer touch-control.
The secret: replace currently used indium tin oxide (ITO) — which is expensive, rare, and worse, brittle — with cheap, flexible metal nanowires that can even be sprayed on.
Unfortunately, there has been no simple way to design a touchscreen using nanowires that will provide an optimum combination of low resistance, evenness, and transparency.
It’s trial-and-error: create a batch with a new wire aspect ratio (length/diameter), density, etc., percolate it (like filtering coffee), and see if the thing works — or just forms a random network with gaps. Frustrating, slow.
Now researchers at the University of Pennsylvania** and Duke University** have developed a clever workaround: computer simulation of various combinations of nanowire length and diameter, the number of nanowires, the area they cover, and contact resistance (at wire connections) to reach the Goldilocks zone: the optimum combination of electrical properties and transparency.
No information on commercialization of this research was available from the team, but future research will focus on nanowire orientation, various continuous deposition methods, variation in nanowire length and diameter, and different processing techniques.
The research was supported by the National Science Foundation and Penn’s Materials Science Research and Engineering Center.
* How a resistive touchscreen works
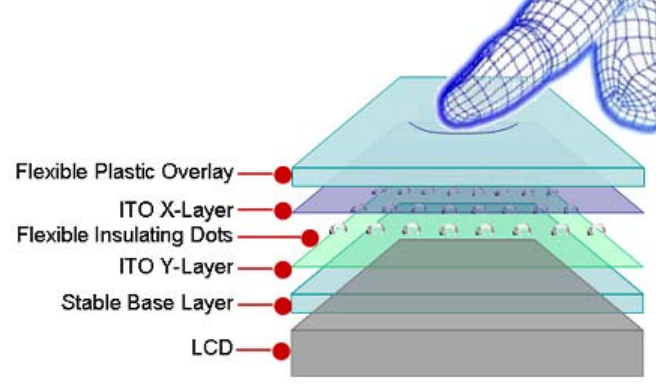
(Credit: Cypress Semiconductor)
Indium tin oxide is deposited as two thin layers on either side of a separator film. When you touch the screen (fingertip or stylus), you change the electrical resistance between the two ITO layers so your phone or monitor can register where you are touching.
More here.
** The research was conducted by graduate student Rose Mutiso, undergraduate Michelle Sherrott and professor Karen Winey, all of the Department of Materials Science and Engineering in Penn’s School of Engineering and Applied Science. They collaborated with graduate student Aaron Rathmell, and professor Benjamin Wiley of Duke’s Department of Chemistry.