How to generate new neurons in brains, spinal cords of living adult mammals
March 4, 2014
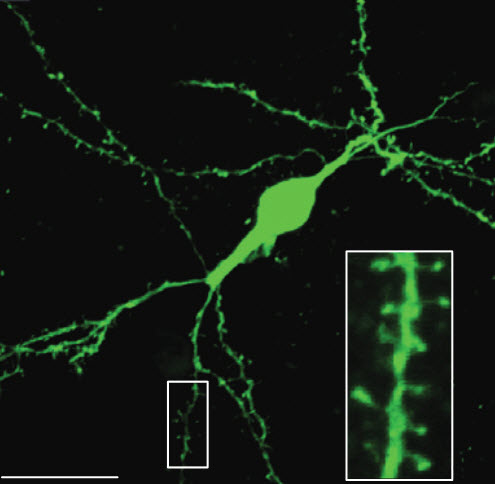
Induced adult neuroblasts generated from astrocytes developed into functionally mature neurons (scale bar, 20 microns) (credit: Zhida Su et al./Nature Communications)
UT Southwestern Medical Center researchers have created new nerve cells in the brains and spinal cords of living mammals without the need for stem cell transplants to replenish lost cells.
In a comment to a KurzweilAI news article, “Brain signals from a primate directly move paralyzed limbs in another primate ‘avatar,'” “Cazbot” wondered if “a sort of ‘neural jumper’ could be used to bridge damaged sections of spinal chord.”
This new research indicates it may someday be possible to do just that — by regenerating neurons from the body’s own cells to repair traumatic brain injury or spinal cord damage, or to treat conditions such as Alzheimer’s disease.
Spinal cord injuries can lead to an irreversible loss of neurons, and along with scarring, can ultimately lead to impaired motor and sensory functions. Scientists are hopeful that regenerating cells can be an avenue to repair damage, but adult spinal cords have limited ability to produce new neurons. Biomedical scientists have transplanted stem cells to replace neurons, but have faced other hurdles, underscoring the need for new methods of replenishing lost cells.
However, the researchers stressed that it is too soon to know whether the neurons created in these initial studies resulted in any functional improvements, a goal for future research.
Reprogramming astrocytes to form neurons
Scientists in UT Southwestern’s Department of Molecular Biology first successfully turned astrocytes — the most common non-neuronal brain cells — into neurons that then formed networks in mice. They have now successfully turned scar-forming astrocytes in the spinal cords of adult mice into neurons. The latest findings are published today in Nature Communications and follow previous findings published in Nature Cell Biology.
“Our earlier work was the first to clearly show in vivo (in a living animal) that mature astrocytes can be reprogrammed to become functional neurons without the need of cell transplantation. The current study did something similar in the spine, turning scar-forming astrocytes into progenitor cells called neuroblasts that regenerated into neurons,” said Chun-Li Zhang, assistant professor of molecular biology at UT Southwestern and senior author of both studies.
“Astrocytes are abundant and widely distributed both in the brain and in the spinal cord. In response to injury, these cells proliferate and contribute to scar formation. Once a scar has formed, it seals the injured area and creates a mechanical and biochemical barrier to neural regeneration,” Zhang explained. “Our results indicate that the astrocytes may be ideal targets for in vivo reprogramming.”
The neurogenesis procedure
The scientists’ two-step approach first introduces a biological substance that regulates the expression of genes, called a transcription factor, into areas of the brain or spinal cord where that factor is not highly expressed in adult mice. Of 12 transcription factors tested, only SOX2 switched fully differentiated, adult astrocytes to an earlier neuronal precursor, or neuroblast, stage of development, Zhang said.
In the second step, the researchers gave the mice a drug called valproic acid (VPA) that encouraged the survival of the neuroblasts and their maturation (differentiation) into neurons. VPA has been used to treat epilepsy for more than half a century and also is prescribed to treat bipolar disorder and to prevent migraine headaches, he said.
The current study reports neurogenesis (neuron creation) occurred in the spinal cords of both adult and aged (over one-year old) mice of both sexes, although the response was much weaker in the aged mice, Dr. Zhang said.
Researchers now are searching for ways to boost the number and speed of neuron creation. Neuroblasts took four weeks to form and eight weeks to mature into neurons, slower than neurogenesis reported in lab dish experiments, so researchers plan to conduct experiments to determine if the slower pace helps the newly generated neurons properly integrate into their environment.
In the spinal cord study, SOX2-induced mature neurons created from reprogramming of astrocytes persisted for 210 days after the start of the experiment, the longest time the researchers examined, he added.
Because tumor growth is a concern when cells are reprogrammed to an earlier stage of development, the researchers followed the mice in the Nature Cell Biology study for nearly a year to look for signs of tumor formation and reported finding none.
Both studies received support from the American Heart Association, the Welch Foundation, the Ellison Medical Foundation, and the National Institutes of Health. The Nature Cell Biology study also received support from an NIH Director’s New Innovator Award and from the Whitehall Foundation.
Abstract of Nature Communications paper
Spinal cord injury (SCI) leads to irreversible neuronal loss and glial scar formation, which ultimately result in persistent neurological dysfunction. Cellular regeneration could be an ideal approach to replenish the lost cells and repair the damage. However, the adult spinal cord has limited ability to produce new neurons. Here we show that resident astrocytes can be converted to doublecortin (DCX)-positive neuroblasts by a single transcription factor, SOX2, in the injured adult spinal cord. Importantly, these induced neuroblasts can mature into synapse-forming neurons in vivo. Neuronal maturation is further promoted by treatment with a histone deacetylase inhibitor, valproic acid (VPA). The results of this study indicate that in situ reprogramming of endogenous astrocytes to neurons might be a potential strategy for cellular regeneration after SCI.
Abstract of Nature Cell Biology paper
Adult differentiated cells can be reprogrammed into pluripotent stem cells or lineage-restricted proliferating precursors in culture; however, this has not been demonstrated in vivo. Here, we show that the single transcription factor SOX2 is sufficient to reprogram resident astrocytes into proliferative neuroblasts in the adult mouse brain. These induced adult neuroblasts (iANBs) persist for months and can be generated even in aged brains. When supplied with BDNF and noggin or when the mice are treated with a histone deacetylase inhibitor, iANBs develop into electrophysiologically mature neurons, which functionally integrate into the local neural network. Our results demonstrate that adult astrocytes exhibit remarkable plasticity in vivo, a feature that might have important implications in regeneration of the central nervous system using endogenous patient-specific glial cells.