Modeling how neurons work together to perform movements: not as random as we thought
June 23, 2014
Multiphoton microscopy of mouse motor neurons (credit: Zeiss Microscopy/flickr)
In a bid to better understand the brain and also to create robotics limbs that behave more realistically, a team of three European universities has developed a highly accurate new model of how neurons behave when performing complex movements.
The results from the University of Cambridge, University of Oxford, and the Ecole Polytechnique Fédérale de Lausanne (EPFL) are published in the June 18 edition of the journal Neuron.
The new theory was inspired by recent experiments carried out at Stanford University, which had uncovered some key aspects of the signals that neurons emit before, during, and after a movement. “There is a remarkable synergy in the activity recorded simultaneously in hundreds of neurons,” said Guillaume Hennequin, PhD, of EPFL’s Department of Engineering, who led the research. “In contrast, previous models of cortical circuit dynamics predict a lot of redundancy, and therefore poorly explain what happens in the motor cortex during movements.”
In addition to helping us better understand the brain, better models of how neurons behave will aid in designing prosthetic limbs controlled via electrodes implanted in the brain. “Our theory could provide a more accurate guess of how neurons would want to signal both movement intention and execution to the robotic limb,” said Hennequin.
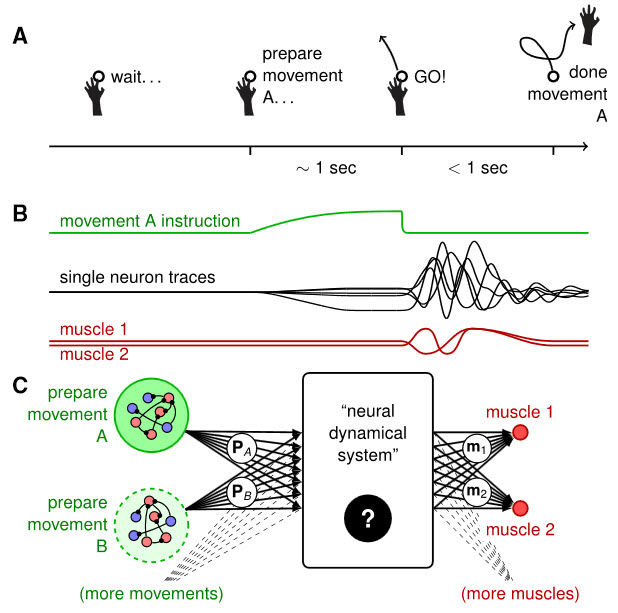
Dynamical Systems View of Movement Planning and Execution. (A) A typical delayed movement generation task starts with the instruction of what movement must be prepared. The arm must then be held still until the go cue is given, upon which the movement is performed. (B) During the preparatory period, model neurons receive a ramp input (green). Following the go cue, that input is withdrawn, leaving the network activity free to evolve from the initial condition set up during the preparatory period. Model neurons then exhibit transient oscillations (black) that drive muscle activity (red). (C) Black-box view on movement generation. Muscles (red, right) are thought to be activated by a population of motor cortical neurons (‘‘neural dynamical system,’’ middle). To prepare the movement, this network is initialized in a desired state by the slow activation of a movement-specific pool of neurons (green, left). (Credit: Guillaume Hennequin et al./Neuron)
Using control theory, the researchers devised a model of neural behavior that achieves a balance between excitatory and inhibitory synaptic signals. The model can accurately reproduce a range of multidimensional movement patterns. The researchers found that neurons in the motor cortex might not be wired together with nearly as much randomness as had been previously thought.
Future directions for the research include building a more realistic, closed-loop model of movement generation in which feedback from the limbs is actively used by the brain to correct for small errors in movement execution.
Abstract of Neuron paper
- Stability-optimized circuits with strong synapses produce complex neural dynamics
- They account for single-cell and population recordings in monkey M1 during reaching
- Their dynamics form a unique basis that can be assembled into complex motor sequences
- They predict a detailed and dynamic excitation/inhibition balance in single neurons
Populations of neurons in motor cortex engage in complex transient dynamics of large amplitude during the execution of limb movements. Traditional network models with stochastically assigned synapses cannot reproduce this behavior. Here we introduce a class of cortical architectures with strong and random excitatory recurrence that is stabilized by intricate, fine-tuned inhibition, optimized from a control theory perspective. Such networks transiently amplify specific activity states and can be used to reliably execute multidimensional movement patterns. Similar to the experimental observations, these transients must be preceded by a steady-state initialization phase from which the network relaxes back into the background state by way of complex internal dynamics. In our networks, excitation and inhibition are as tightly balanced as recently reported in experiments across several brain areas, suggesting inhibitory control of complex excitatory recurrence as a generic organizational principle in cortex.