Molecular switch that controls neuron communication discovered
November 29, 2013
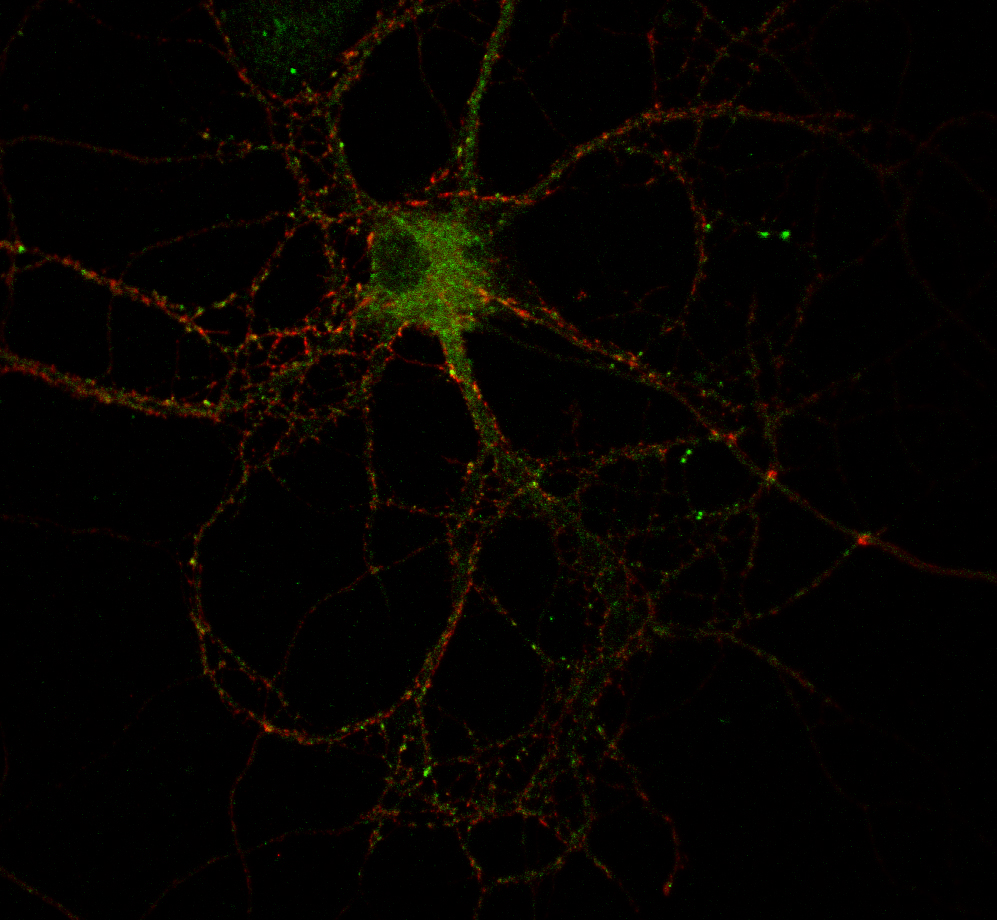
Molecular switch: an image of a hippocampal neuron stained for endogenous RIM1 and SUMO. RIM1 is in red and SUMO in green. (Credit: University of Bristol)
University of Bristol researchers are a step closer to understanding how some of the brain’s 100 billion nerve cells coordinate communications.
Defects in this communication are associated with disorders such as epilepsy, autism and schizophrenia, and therefore these findings could lead to the development of novel neurological therapies.
Background
Neurons in the brain communicate with each other using chemicals called neurotransmitters. This release of neurotransmitter from neurons is tightly controlled by many different proteins inside the neuron. These proteins interact with each other to ensure that neurotransmitter is only released when necessary.
Although the mechanisms that control this release have been extensively studied, the processes that coordinate how and when the component proteins interact is not fully understood.
A molecular switch
Bristol School of Biochemistry researchers have now discovered that one of these proteins called “RIM1α” is modified by a peptide (small protein) named “SUMO” (small ubiquitin-like modifier), which attaches to a specific region in RIM1α. This process acts as a “molecular switch” required for normal neurotransmitter release.
“This research builds on several decades of research identifying key proteins involved in neurotransmitter release,” Dr. Tim Craig, a co-author of the paper, explained to KurzweilAI. “Many of these proteins have multiple roles in the presynapse, but it is unknown how these different roles are regulated. Our research identifies a molecular switch (SUMOylation) that may play a key role in regulating the functions of these proteins.
“The research builds on the team’s earlier work that identified a group of proteins in the brain responsible for protecting nerve cells from damage. It could be used in future for therapies for stroke and other brain diseases. It will be also be useful for in silico [computational] modeling of neurons and synapses. And it provides a potential drug target, as the pathways which we have shown to be regulated by SUMOylation are involved in the pathology of epilepsy and schizophrenia.”
The study is published in the journal Cell Reports (open access). It was funded by the European Research Council [ERC], the Biotechnology and Biological Sciences Research Council [BBSRC] and the Medical Research Council [MRC].
Abstract of Cell Reports paper
The rapid, activity-dependent quantal presynaptic release of neurotransmitter is vital for brain function. The complex process of vesicle priming, fusion, and retrieval is very precisely controlled and requires the spatiotemporal coordination of multiple protein-protein interactions. Here, we show that posttranslational modification of the active zone protein Rab3-interacting molecule 1α (RIM1α) by the small ubiquitin-like modifier 1 (SUMO-1) functions as a molecular switch to direct these interactions and is essential for fast synaptic vesicle exocytosis. RIM1α SUMOylation at lysine residue K502 facilitates the clustering of CaV2.1 calcium channels and enhances the Ca2+ influx necessary for vesicular release, whereas non-SUMOylated RIM1α participates in the docking/priming of synaptic vesicles and maintenance of active zone structure. These results demonstrate that SUMOylation of RIM1α is a key determinant of rapid, synchronous neurotransmitter release, and the SUMO-mediated “switching” of RIM1α between binding proteins provides insight into the mechanisms underpinning synaptic function and dysfunction.