Nanoarray sniffs out and distinguishes ‘breathprints’ of multiple diseases
December 23, 2016
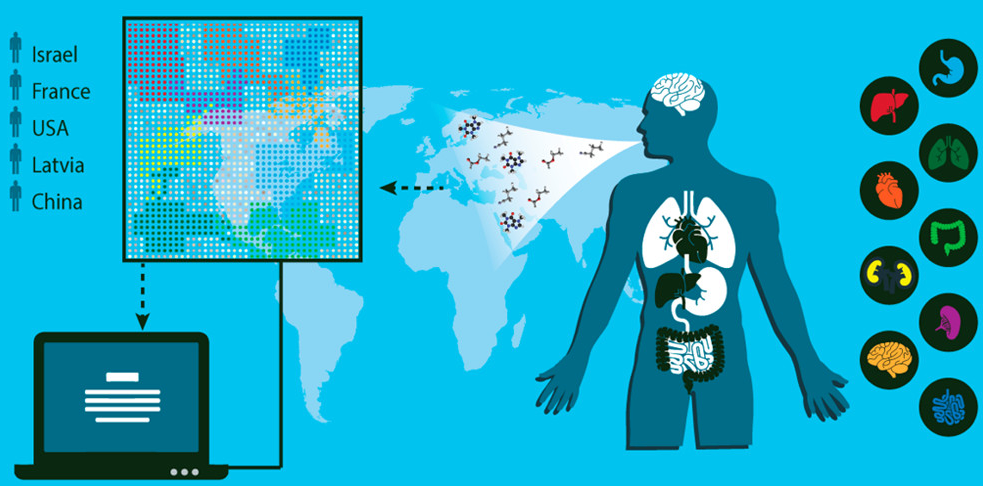
Schematic representation of the concept and design of the study, which involved collection of breath samples from 1404 patients diagnosed with one of 17 different diseases. One breath sample obtained from each subject was analyzed with the artificially intelligent nanoarray for disease diagnosis and classification (represented by patterns in the illustration), and a second sample was analyzed with gas chromatography–mass spectrometry to explore its chemical composition. (credit: Morad K. Nakhleh et al./ACS Nano)
An international team of 63 scientists in 14 clinical departments have identified a unique “breathprint” for 17 diseases with 86% accuracy and have designed a noninvasive, inexpensive, and miniaturized portable device that screens breath samples to classify and diagnose several types of diseases, they report in an open-access paper in the journal ACS Nano.
As far back as around 400 B.C., doctors diagnosed some diseases by smelling a patient’s exhaled breath, which contains nitrogen, carbon dioxide, oxygen, and a small amount of more than 100 other volatile chemical components. Relative amounts of these substances vary depending on the state of a person’s health. For example, diabetes creates a sweet breath smell. More recently, several teams of scientists have developed experimental breath analyzers, but most of these instruments focus on one disease, such as diabetes and melanoma, or a few diseases.
Detecting 17 diseases
The researchers developed an array of nanoscale sensors to detect the individual components in thousands of breath samples collected from 1404 patients who were either healthy or had one of 17 different diseases*, such as kidney cancer or Parkinson’s disease.
The team used mass spectrometry to identify the breath components associated with each disease. By analyzing the results with artificial intelligence techniques (binary classifiers), the team found that each disease produces a unique breathprint, based on differing amounts of 13 volatile organic chemical (VOC) components. They also showed that the presence of one disease would not prevent the detection of others — a prerequisite for developing a practical device to screen and diagnose various diseases.
Based on the research, the team designed an organic layer that functions as a sensing layer (recognition element) for adsorbed VOCs and an electrically conductive nanoarray based on resistive layers of molecularly modified gold nanoparticles and a random network of single-wall carbon nanotubes. The nanoparticles and nanotubes have different electrical conductivity patterns associated with different diseases.**
The authors received funding from the ERC and LCAOS of the European Union’s Seventh Framework Programme for Research and Technological Development, the EuroNanoMed Program under VOLGACORE, and the Latvian Council of Science.
* Lung cancer, colorectal cancer, head and neck cancer, ovarian cancer, bladder cancer, prostate cancer, kidney cancer, gastric cancer, Crohn’s disease, ulcerative colitis, irritable bowel syndrome, idiopathic Parkinson’s, atypical Parkinsonism, multiple sclerosis, pulmonary arterial hypertension, pre-eclampsia, and chronic kidney disease.
** During exposure to breath samples, interaction between the VOC components and the organic sensing layer changes the electrical resistance of the sensors. The relative change of sensor’s resistance at the peak (beginning), middle, and end of the exposure, as well as the area under the curve of the whole measurement were measured. All breath samples identified by the AI nanoarray were also examined using an independent lab-based analytical technique: gas chromatography linked with mass spectrometry.
Abstract of Diagnosis and Classification of 17 Diseases from 1404 Subjects via Pattern Analysis of Exhaled Molecules
We report on an artificially intelligent nanoarray based on molecularly modified gold nanoparticles and a random network of single-walled carbon nanotubes for noninvasive diagnosis and classification of a number of diseases from exhaled breath. The performance of this artificially intelligent nanoarray was clinically assessed on breath samples collected from 1404 subjects having one of 17 different disease conditions included in the study or having no evidence of any disease (healthy controls). Blind experiments showed that 86% accuracy could be achieved with the artificially intelligent nanoarray, allowing both detection and discrimination between the different disease conditions examined. Analysis of the artificially intelligent nanoarray also showed that each disease has its own unique breathprint, and that the presence of one disease would not screen out others. Cluster analysis showed a reasonable classification power of diseases from the same categories. The effect of confounding clinical and environmental factors on the performance of the nanoarray did not significantly alter the obtained results. The diagnosis and classification power of the nanoarray was also validated by an independent analytical technique, i.e., gas chromatography linked with mass spectrometry. This analysis found that 13 exhaled chemical species, called volatile organic compounds, are associated with certain diseases, and the composition of this assembly of volatile organic compounds differs from one disease to another. Overall, these findings could contribute to one of the most important criteria for successful health intervention in the modern era, viz. easy-to-use, inexpensive (affordable), and miniaturized tools that could also be used for personalized screening, diagnosis, and follow-up of a number of diseases, which can clearly be extended by further development.