Nanoscale probes light up when they detect specific diseases
November 22, 2012

Nanostructures called BRIGHTs seek out biomarkers on cells and then beam brightly to reveal their locations. In the tiny gap between the gold skin and the gold core of the cleaved BRIGHT (visible to the upper left), there is an electromagnetic hot spot that lights up the reporter molecules trapped there. (Credit: Naveen Gandra/Washington University in St. Louis)
Washington University researchers have developed nanoscale probes that bind to biomarkers of disease and, when hit by an infrared laser, light up to reveal their location.
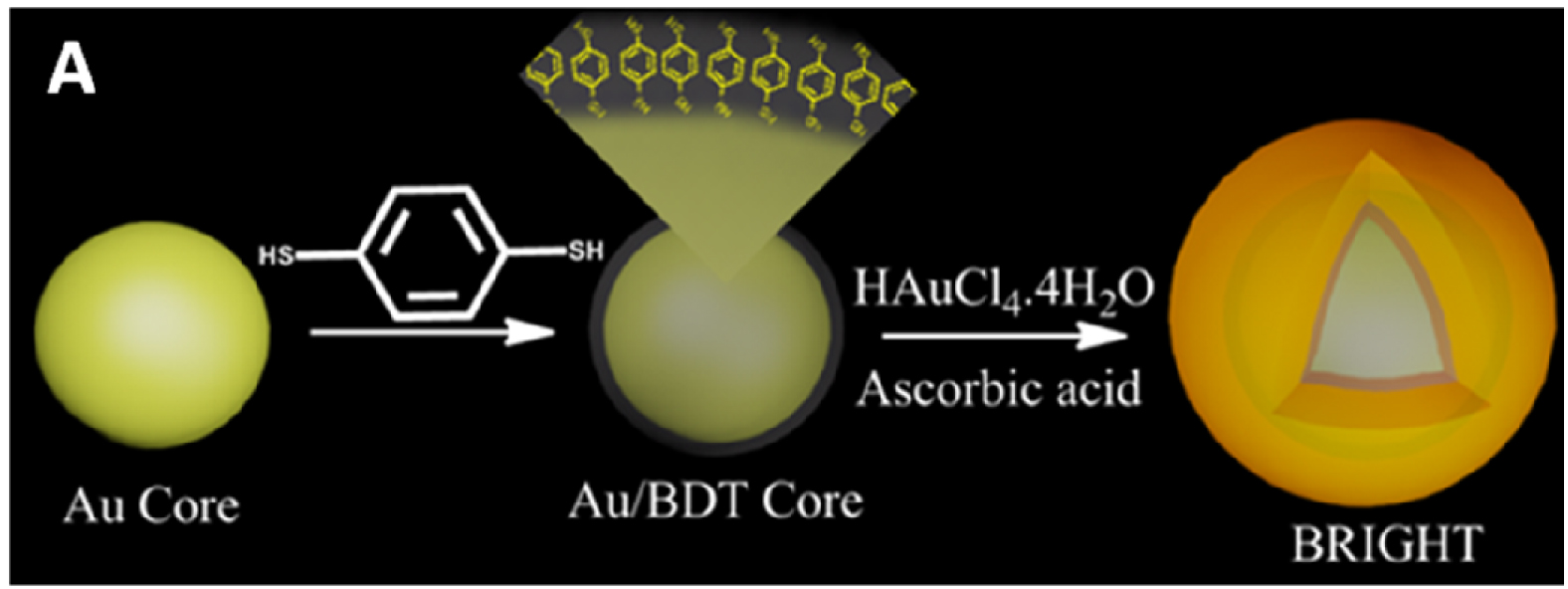
The probes, called BRIGHTs (Bi-layered Raman-Intense Gold nanostructures with Hidden Tags), comprise 20 nm. diameter gold nanoparticles covered with molecules called Raman reporters, which are in turn covered by a thin shell of gold that spontaneously forms a dodecahedron (a polyhedron with 12 regular pentagonal faces).
Raman reporters are molecules whose atoms respond to a probe laser by scattering light at characteristic wavelengths. The shell and core create an electromagnetic hotspot (concentrated energy) in the gap between them that boosts the reporters’ emission by a factor of nearly a trillion.
BRIGHTs shine about one hundred billion times more brightly than isolated Raman reporters and about 20 times more intensely than the next-closest competitor probe, says Srikanth Singamaneni, PhD, assistant professor of mechanical engineering and materials science in the School of Engineering & Applied Science at Washington University.

SEM image of BRIGHTS (credit: Naveen Gandra/Washington University in St. Louis)
Singamaneni’s lab has worked for years with Raman spectroscopy, a spectroscopic technique that is used to study the vibrational modes (bending and stretching) of molecules.
Laser light interacts with these modes and the molecule then emits light at higher or lower wavelengths that are characteristic of the molecule.
Spontaneous Raman scattering, as this phenomenon is called, is by nature very weak, but 30 years ago scientists accidently stumbled on the fact that it is much stronger if the molecules are adsorbed on roughened metallic surfaces. Then they discovered that molecules attached to metallic nanoparticles shine even brighter than those attached to rough surfaces.
The intensity boost from surface-enhanced Raman scattering, or SERS, is potentially huge. “It’s well-known that if you sandwich Raman reporters between two plasmonic materials, such as gold or silver, you are going to see dramatic Raman enhancement,” Singamaneni says.
BRIGHTs diagram (credit: Naveen Gandra/Washington University in St. Louis)
Since different Raman reporter molecules respond at different wavelengths, Singamaneni says, it should be possible to design BRIGHTS targeted to different biomolecules that also have different Raman reporters and then monitor them all simultaneously with the same light probe.
And he and Gandra would like to combine BRIGHTS with a drug container, so that the containers could be tracked in the body; the drug would be released only when it reached the target tissue, thus avoiding many of the side effects patients dread.