Neurons made from stem cells drive brain activity after transplantation
November 21, 2012
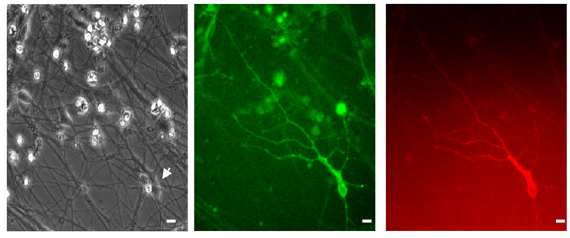
Stem-cell derived neurons carrying genes encoding light-gated conductances (credit: J. C. Pina-Crespo et al./Journal of Neuroscience)
Researchers at Sanford-Burnham Medical Research Institute have found a way to stimulate stem cell-derived neurons to direct cognitive function after transplantation to an existing neural network by using optogenetic stimulation — getting us a step closer to using these cells to treat Alzheimer’s disease and other neurodegenerative conditions.
Researchers and patients look forward to the day when stem cells might be used to replace dying brain cells in Alzheimer’s disease and other neurodegenerative conditions.
Scientists are currently able to make neurons and other brain cells from stem cells, but getting these neurons to properly function when transplanted to the host has proven to be more difficult.
“We showed for the first time that embryonic stem cells that we’ve programmed to become neurons can integrate into existing brain circuits and fire patterns of electrical activity that are critical for consciousness and neural network activity,” said Stuart A. Lipton, M.D., Ph.D., senior author of the study. Lipton is director of Sanford-Burnham’s Del E. Webb Neuroscience, Aging, and Stem Cell Research Center and a clinical neurologist.
Lipton and his team — including colleagues at Sanford-Burnham and Stanford University — transplanted human stem cell-derived neurons into a rodent hippocampus, the brain’s information-processing center.
Then they specifically activated the transplanted neurons with optogenetic stimulation, a relatively new technique that combines light and genetics to precisely control cellular behavior in living tissues or animals.
To determine if the newly transplanted, light-stimulated human neurons were actually working, Lipton and his team measured high-frequency oscillations in existing neurons at a distance from the transplanted ones. They found that the transplanted neurons triggered the existing neurons to fire high-frequency oscillations. Faster neuronal oscillations are usually better — they’re associated with enhanced performance in sensory-motor and cognitive tasks.
The transplanted human neurons not only conducted electrical impulses, they also roused neighboring neuronal networks into firing — at roughly the same rate they would in a normal, functioning hippocampus.
The therapeutic outlook for this technology looks promising. “Based on these results, we might be able to restore brain activity — and thus restore motor and cognitive function — by transplanting easily manipulated neuronal cells derived from embryonic stem cells,” Lipton said.
This research was funded by the California Institute for Regenerative Medicine and the U.S. National Institutes of Health, National Institute of Environmental Health Sciences, National Institute of Neurological Disorders and Stroke, and the National Eye Institute.