New device sees bacteria behind the eardrum
May 31, 2012
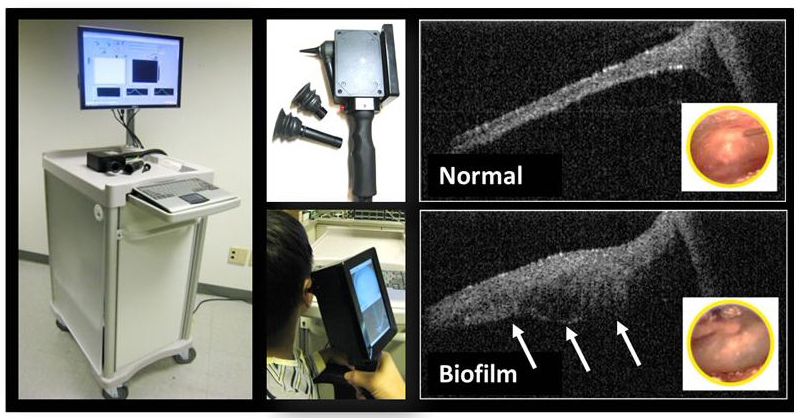
University of Illinois researchers tested a prototype of a new device that can see biofilms behind the eardrum to better diagnose and treat chronic ear infections (credit: Stephen Boppart)
Doctors can now get a peek behind the eardrum to better diagnose and treat chronic ear infections, thanks to a new medical imaging device invented by University of Illinois researchers. The device could usher in a new suite of non-invasive, 3-D diagnostic imaging tools for primary-care physicians, including examination of eyes, mouth, nose, and skin.
Ear infections are the most common conditions that pediatricians treat. Chronic ear infections can damage hearing and often require surgery to place drainage tubes in the eardrum, and problems can persist into adulthood.
Studies have found that patients who suffer from chronic ear infections may have a film of bacteria or other microorganisms that builds up behind the eardrum, very similar to dental plaque on unbrushed teeth. Finding and monitoring these so-called biofilms are important for successfully identifying and treating chronic ear infections.
“We know that antibiotics don’t always work well if you have a biofilm, because the bacteria protect themselves and become resistant,” said University of Illinois electrical and computer engineering professor Stephen Boppart. “In the presence of a chronic ear infection that has a biofilm, the bacteria may not respond to the usual antibiotics, and you need to stop them. But without being able to detect the biofilm, we have no idea whether or not it’s responding to treatment.”
Moreover, middle-ear biofilms are difficult to diagnose. A doctor looking through a standard otoscope sees only the eardrum’s surface, not the bacteria-seeded biofilm lurking behind it waiting to bloom into infection. Invasive tests can provide evidence of a biofilm, but are unpleasant for the patient and cannot be used routinely.
Optical coherence tomography
The new device is an application of a technique called optical coherence tomography (OCT), a non-invasive imaging system devised by Boppart’s group. It uses beams of light to collect high-resolution, 3D tissue images, scanning through the eardrum to the biofilm behind it — much like ultrasound imaging, but using light.
“We send the light into the ear canal, and it scatters and reflects from the tympanic membrane and the biofilm behind it,” said graduate student Cac Nguyen, the lead author of the paper. “We measure the reflection, and with the reference light we can get the structure in depth.”
The single scan is performed in a fraction of a second — speed is a necessity for images a few millimeters deep behind the eardrum. Thus, doctors can see not only the presence of a biofilm, but also how thick it is and its position against the eardrum.
The paper marks the first demonstration of using the ear OCT device to detect biofilms in human patients. To test their device, the researchers worked with clinicians at Carle Foundation Hospital in Urbana, Ill., to scan patients with diagnosed chronic ear infections, as well as patients with normal ears. The device identified biofilms in all patients with chronic infections, while none of the normal ears showed evidence of biofilms.
Next, the researchers plan to investigate different ear pathology, particularly comparing acute and chronic infections, and will examine the relationship between biofilms and hearing loss. They hope that improved diagnostics will lead to better treatment and referral practices.
They hope to make their device — currently a hand-held prototype — even more compact, easy to use, and low-cost. The ear-imaging device is the first in a suite of OCT-based imaging tools that the group plans to develop.
Doctors could change the tip of the new OCT device, for example, to look at the eyes, mouth, nose, or skin.
Ref.: Cac T. Nguyen et al., Noninvasive in vivo optical detection of biofilm in the human middle ear, Proceedings of the National Academy of Sciences, 2012, DOI: 10.1073/pnas.1201592109