New drug reverses effects of Alzheimer’s disease in mice
August 7, 2014
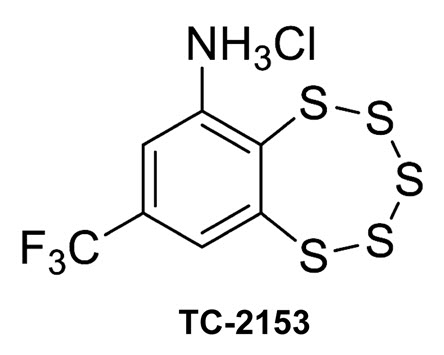
TC-2153, a novel inhibitor of the STEP protein (credit: Jian Xu et al./PLoS Biology)
Researchers at Yale School of Medicine have discovered a new drug compound that reverses the brain deficits of Alzheimer’s disease in mice. Their findings are published in the Aug. 5 issue of the journal PLoS Biology (open access).
The compound, TC-2153, inhibits the negative effects of a protein called STtriatal-Enriched tyrosine Phosphatase (STEP) on learning and memory. These cognitive functions are impaired in Alzheimer’s.
“Decreasing STEP levels reversed the effects of Alzheimer’s disease in mice,” said lead author Paul Lombroso, M.D., professor in the Yale Child Study Center and in the Departments of Neurobiology and Psychiatry at Yale School of Medicine.
Lombroso and co-authors studied thousands of small molecules, searching for those that would inhibit STEP activity. Once identified, those STEP-inhibiting compounds were tested in brain cells in the lab to examine how effectively they could halt the effects of STEP. They examined the most promising compound in a mouse model of Alzheimer’s disease, and found a reversal of deficits in several cognitive exercises that gauged the animals’ ability to remember previously seen objects.
High levels of STEP proteins keep synapses in the brain from strengthening. Synaptic strengthening is a process that is required for people to turn short-term memories into long-term memories. When STEP is elevated in the brain, it depletes receptors from synaptic sites, and inactivates other proteins that are necessary for proper cognitive function. This disruption can result in Alzheimer’s disease or a number of neuropsychiatric and neurodegenerative disorders, all marked by cognitive deficits.
“The small molecule inhibitor is the result of a five-year collaborative effort to search for STEP inhibitors,” said Lombroso. “A single dose of the drug results in improved cognitive function in mice. Animals treated with TC compound were indistinguishable from a control group in several cognitive tasks.”
The team is currently testing the TC compound in other animals with cognitive defects, including rats and non-human primates. “These studies will determine whether the compound can improve cognitive deficits in other animal models,” said Lombroso. “Successful results will bring us a step closer to testing a drug that improves cognition in humans.”
The study was funded by the National Institute of Health, the American Health Assistance Foundation, and the Alzheimer’s Drug Discovery Foundation.
Abstract of PLoS Biology paper
STEP (STriatal-Enriched protein tyrosine Phosphatase) is a neuron-specific phosphatase that regulates N-methyl-D-aspartate receptor (NMDAR) and α-amino-3-hydroxy-5-methyl-4-isoxazolepropionicacid receptor (AMPAR) trafficking, as well as ERK1/2, p38, Fyn, and Pyk2 activity. STEP is overactive in several neuropsychiatric and neurodegenerative disorders, including Alzheimer’s disease (AD). The increase in STEP activity likely disrupts synaptic function and contributes to the cognitive deficits in AD. AD mice lacking STEP have restored levels of glutamate receptors on synaptosomal membranes and improved cognitive function, results that suggest STEP as a novel therapeutic target for AD. Here we describe the first large-scale effort to identify and characterize small-molecule STEP inhibitors. We identified the benzopentathiepin 8-(trifluoromethyl)-1,2,3,4,5-benzopentathiepin-6-aminehydrochloride (known as TC-2153) as an inhibitor of STEP with an IC50 of 24.6 nM. TC-2153 represents a novel class of PTP inhibitors based upon a cyclic polysulfide pharmacophore that forms a reversible covalent bond with the catalytic cysteine in STEP. In cell-based secondary assays, TC-2153 increased tyrosine phosphorylation of STEP substrates ERK1/2, Pyk2, and GluN2B, and exhibited no toxicity in cortical cultures. Validation and specificity experiments performed in wild-type (WT) and STEP knockout (KO) cortical cells and in vivo in WT and STEP KO mice suggest specificity of inhibitors towards STEP compared to highly homologous tyrosine phosphatases. Furthermore, TC-2153 improved cognitive function in several cognitive tasks in 6- and 12-mo-old triple transgenic AD (3xTg-AD) mice, with no change in beta amyloid and phospho-tau levels.