New multilayer graphene structure allows ‘ultraprecise,’ ‘ultrafast’ water filtering
February 18, 2014
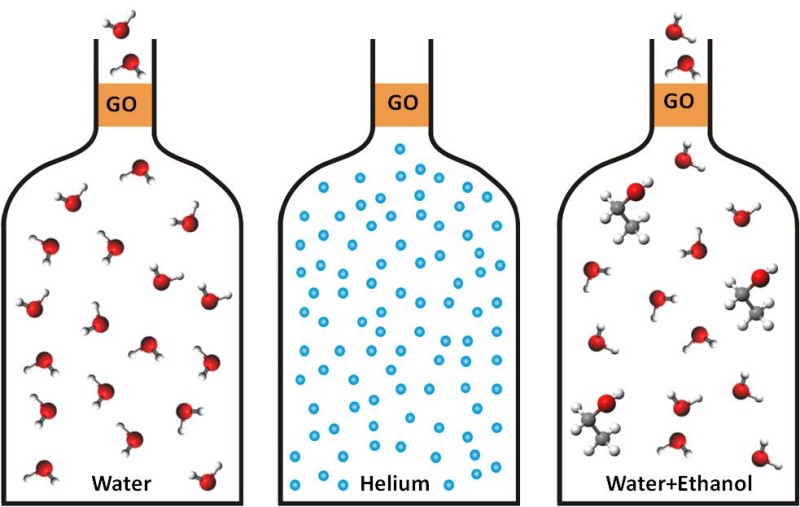
Water easily evaporates through graphene-oxide membranes but they represent an impermeable barrier for other molecules (credit: Rahul R.Nair)
University of Manchester researchers have taken another key step toward a seawater filter: they’ve developed one-atom-wide graphene-oxide (GO) capillaries by building multilayer GO membranes (laminates).
As described in Science, these new laminates allow for “ultraprecise” selection of molecules that can go through the filter and “ultrafast” flow of water.
The new GO filters have an “astonishingly” accurate mesh that allows them to distinguish between atomic species that are only a few percent different in size. The filters block passage of ions (charged atoms) that are larger than 9 angstroms (.9 nanometers).
“The water filtration is as fast and as precise as one could possibly hope for such narrow capillaries,” said study co-author Rahul Nair. “Now we want to control the graphene mesh size and reduce it below nine Angstroms to filter out even the smallest salts like in seawater. Our work shows that it is possible.”
“Our ultimate goal is to make a filter device that allows a glass of drinkable water made from seawater after a few minutes of hand pumping,” added study co-author Irina Grigorieva. “We are not there yet but this is no longer science fiction.”
Technical details
GO is hydrophobic — it repels water — but narrow capillaries made from graphene vigorously suck in water allowing its rapid permeation, if the water layer is only one atom thick — that is, as thin as graphene itself.
This bizarre property has attracted intense academic and industrial interest with intent to develop new water filtration and desalination technologies.
One-atom-wide GO capillaries can now be made easily and cheaply by piling layers of GO on top of each other. The resulting multilayer stacks (laminates) have a structure similar to nacre (mother of pearl), which makes them also mechanically strong.
Two years ago, the researchers discovered that thin membranes made from such laminates were impermeable to all gases and vapors, except for water. This means that even helium, the hardest gas to block off, cannot pass through the membranes, whereas water vapor went through with no resistance.
The researchers report that, if immersed in water, the laminates become slightly swollen but still allow ultrafast flow of not one but two monolayers of water.
The work was done in collaboration with a group led by Hengan Wu from the Chinese Academy of Sciences, who carried out extensive computer simulations to understand the filtration mechanism.
Abstract of Science paper
Graphene-based materials can have well-defined nanometer pores and can exhibit low frictional water flow inside them, making their properties of interest for filtration and separation. We investigate permeation through micrometer-thick laminates prepared by means of vacuum filtration of graphene oxide suspensions. The laminates are vacuum-tight in the dry state but, if immersed in water, act as molecular sieves, blocking all solutes with hydrated radii larger than 4.5 angstroms. Smaller ions permeate through the membranes at rates thousands of times faster than what is expected for simple diffusion. We believe that this behavior is caused by a network of nanocapillaries that open up in the hydrated state and accept only species that fit in. The anomalously fast permeation is attributed to a capillary-like high pressure acting on ions inside graphene capillaries.