New optical microscopy technique unravels role of ‘oxidative stress’ in neural tissue
April 24, 2014
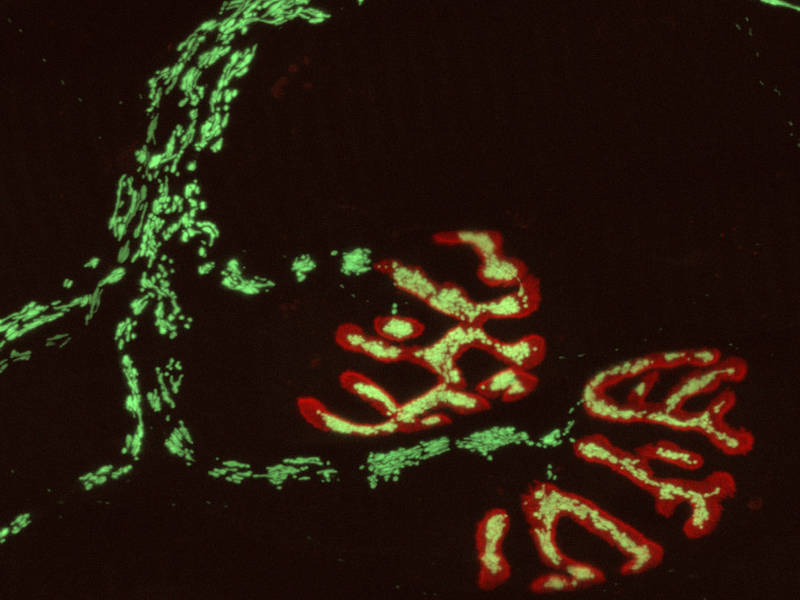
The micrograph shows a peripheral nerve, with the neuromuscular endplates stained in red. The nerve-cell mitochondria were imaged with a fluorescent redox sensor (green in the cytoplasm, yellow at the endplates). (Credit: M. Kerschensteiner and T. Misgeld)
Scientists at the Technische Universität München (TUM) and the Ludwig-Maximilians-Universität München (LMU) have developed a new optical microscopy technique to unravel the role of “oxidative stress” in healthy as well as injured nervous systems. The work is reported in the latest issue of Nature Medicine.
Reactive oxygen species are important intracellular signaling molecules, but their mode of action is complex. In low concentrations they regulate key aspects of cellular function and behavior, while at high concentrations they can cause “oxidative stress,” which damages organelles, membranes and DNA.
“Our new optical approach allows us to visualize the redox state of important cellular organelles, mitochondria, in real time in living tissue,” says LMU Professor Martin Kerschensteiner. Mitochondria are the cell’s power plants, which convert nutrients into usable energy.
In earlier studies, Kerschensteiner and and TUM Professor Thomas Misgeld obtained evidence that oxidative damage of mitochondria might contribute to the destruction of axons in inflammatory diseases such as multiple sclerosis.
Precise analysis of oxidation states of individual mitochondria
The new method allows them to record the oxidation states of individual mitochondria with high spatial and temporal resolution. “Redox signals have important physiological functions, but can also cause damage, for example when present in high concentrations around immune cells.”
Kerschensteiner and Misgeld used redox-sensitive variants of the Green Fluorescent Protein (GFP) as visualization tools. “By combining these with other biosensors and vital dyes, we were able to establish an approach that permits us to simultaneously monitor redox signals together with mitochondrial calcium currents, as well as changes in the electrical potential and the proton (pH) gradient across the mitochondrial membrane,” says Thomas Misgeld.
The researchers have applied the technique to two experimental models, and have arrived at some unexpected insights. They can now study redox signal induction in response to neural damage — in this case, spinal cord injury — in the mammalian nervous system. The observations revealed that severance of an axon results in a wave of oxidation of the mitochondria, which begins at the site of damage and is propagated along the fiber. Also, an influx of calcium at the site of axonal resection was shown to be essential for the ensuing functional damage to mitochondria.
Perhaps the most surprising outcome of the new study was that the study’s first author, graduate student Michael Breckwoldt, was able to image, also for the first time, spontaneous contractions of mitochondria that are accompanied by a rapid shift in the redox state of the organelle.
As Misgeld explains, “This appears to be a fail-safe system that is activated in response to stress and temporarily attenuates mitochondrial activity. Under pathological conditions, the contractions are more prolonged and may become irreversible, and this can ultimately result in irreparable damage to the nerve process.”
Abstract of Nature Medicine paper
Mitochondrial redox signals have a central role in neuronal physiology and disease. Here we describe a new optical approach to measure fast redox signals with single-organelle resolution in living mice that express genetically encoded redox biosensors in their neuronal mitochondria. Moreover, we demonstrate how parallel measurements with several biosensors can integrate these redox signals into a comprehensive characterization of mitochondrial function. This approach revealed that axonal mitochondria undergo spontaneous ‘contractions’ that are accompanied by reversible redox changes. These contractions are amplified by neuronal activity and acute or chronic neuronal insults. Multiparametric imaging reveals that contractions constitute respiratory chain–dependent episodes of depolarization coinciding with matrix alkalinization, followed by uncoupling. In contrast, permanent mitochondrial damage after spinal cord injury depends on calcium influx and mitochondrial permeability transition. Thus, our approach allows us to identify heterogeneity among physiological and pathological redox signals, correlate such signals to functional and structural organelle dynamics and dissect the underlying mechanisms.