Quadriplegia patient uses brain-computer interface to move his arm by just thinking
April 26, 2017
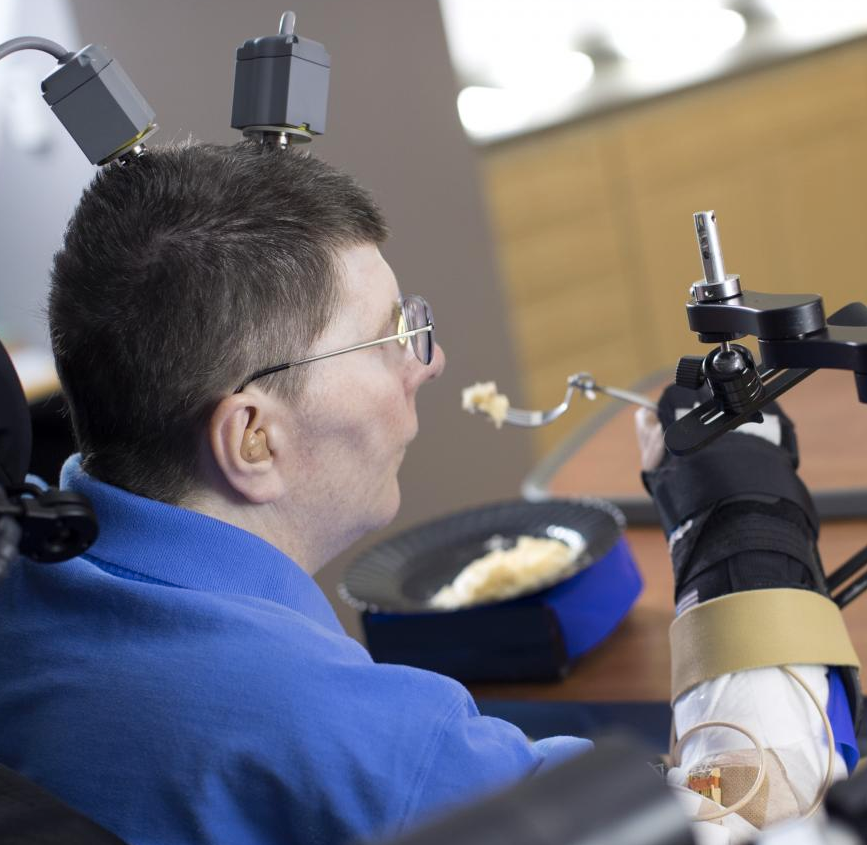
Bill Kochevar, who was paralyzed below his shoulders in a bicycling accident eight years ago, is the first person with quadriplegia to have arm and hand movements restored without robot help (credit: Case Western Reserve University/Cleveland FES Center)
A research team led by Case Western Reserve University has developed the first implanted brain-recording and muscle-stimulating system to restore arm and hand movements for quadriplegic patients.*
In a proof-of-concept experiment, the system included a brain-computer interface with recording electrodes implanted under his skull and a functional electrical stimulation (FES) system that activated his arm and hand — reconnecting his brain to paralyzed muscles.
The research was part of the ongoing BrainGate2 pilot clinical trial being conducted by a consortium of academic and other institutions to assess the safety and feasibility of the implanted brain-computer interface (BCI) system in people with paralysis. Previous Braingate designs required a robot arm.

In 2012 research, Jan Scheuermann, who has quadriplegia, was able to feed herself using a brain-machine interface and a computer-driven robot arm (credit: UPMC)
Kochevar’s eight years of muscle atrophy first required rehabilitation. The researchers exercised Kochevar’s arm and hand with cyclical electrical stimulation patterns. Over 45 weeks, his strength, range of motion. and endurance improved. As he practiced movements, the researchers adjusted stimulation patterns to further his abilities.
To prepare him to use his arm again, Kochevar learned how to use his own brain signals to move a virtual-reality arm on a computer screen. The team then implanted the FES systems’ 36 electrodes that animate muscles in the upper and lower arm, allowing him to move the actual arm.
Kochevar can now make each joint in his right arm move individually. Or, just by thinking about a task such as feeding himself or getting a drink, the muscles are activated in a coordinated fashion.
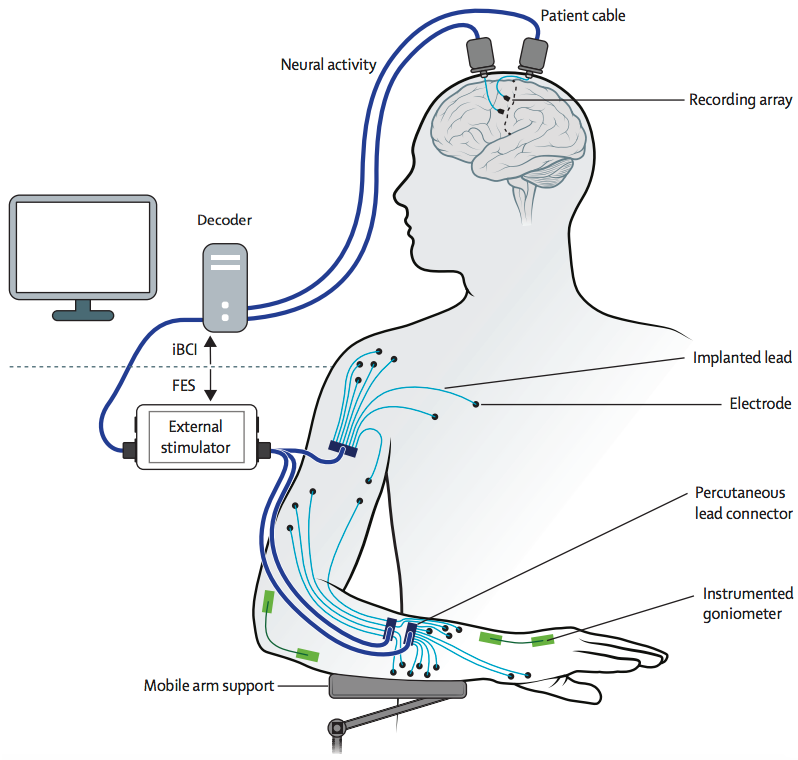
Neural activity (generated when Kochevar imagines movement of his arm and hand) is recorded from two 96-channel microelectrode arrays implanted in the motor cortex, on the surface of the brain. The implanted brain-computer interface translates the recorded brain signals into specific command signals that determine the amount of stimulation to be applied to each functional electrical stimulation (FES) electrode in the hand, wrist, arm, elbow and shoulder, and to a mobile arm support. (credit: A Bolu Ajiboye et al./The Lancet)
“Our research is at an early stage, but we believe that this neuro-prosthesis could offer individuals with paralysis the possibility of regaining arm and hand functions to perform day-to-day activities, offering them greater independence,” said lead author Dr Bolu Ajiboye, Case Western Reserve University. “So far, it has helped a man with tetraplegia to reach and grasp, meaning he could feed himself and drink. With further development, we believe the technology could give more accurate control, allowing a wider range of actions, which could begin to transform the lives of people living with paralysis.”
Work is underway to make the brain implant wireless, and the investigators are improving decoding and stimulation patterns needed to make movements more precise. Fully implantable FES systems have already been developed and are also being tested in separate clinical research.
A study of the work was published in the The Lancet March 28, 2017.
Writing in a linked Comment to The Lancet, Steve Perlmutter, M.D., University of Washington, said: “The goal is futuristic: a paralysed individual thinks about moving her arm as if her brain and muscles were not disconnected, and implanted technology seamlessly executes the desired movement… This study is groundbreaking as the first report of a person executing functional, multi-joint movements of a paralysed limb with a motor neuro-prosthesis. However, this treatment is not nearly ready for use outside the lab. The movements were rough and slow and required continuous visual feedback, as is the case for most available brain-machine interfaces, and had restricted range due to the use of a motorised device to assist shoulder movements… Thus, the study is a proof-of-principle demonstration of what is possible, rather than a fundamental advance in neuro-prosthetic concepts or technology. But it is an exciting demonstration nonetheless, and the future of motor neuro-prosthetics to overcome paralysis is brighter.”
* The study was funded by the US National Institutes of Health and the US Department of Veterans Affairs. It was conducted by scientists from Case Western Reserve University, Department of Veterans Affairs Medical Center, University Hospitals Cleveland Medical Center, MetroHealth Medical Center, Brown University, Massachusetts General Hospital, Harvard Medical School, Wyss Center for Bio and Neuroengineering. The investigational BrainGate technology was initially developed in the Brown University laboratory of John Donoghue, now the founding director of the Wyss Center for Bio and Neuroengineering in Geneva, Switzerland. The implanted recording electrodes are known as the Utah array, originally designed by Richard Normann, Emeritus Distinguished Professor of Bioengineering at the University of Utah. The report in Lancet is the result of a long-running collaboration between Kirsch, Ajiboye and the multi-institutional BrainGate consortium. Leigh Hochberg, a neurologist and neuroengineer at Massachusetts General Hospital, Brown University and the VA RR&D Center for Neurorestoration and Neurotechnology in Providence, Rhode Island, directs the pilot clinical trial of the BrainGate system and is a study co-author.
Case | Man with quadriplegia employs injury bridging technologies to move again – just by thinking
Abstract of Restoration of reaching and grasping movements through brain-controlled muscle stimulation in a person with tetraplegia: a proof-of-concept demonstration
Background: People with chronic tetraplegia, due to high-cervical spinal cord injury, can regain limb movements through coordinated electrical stimulation of peripheral muscles and nerves, known as functional electrical stimulation (FES). Users typically command FES systems through other preserved, but unrelated and limited in number, volitional movements (eg, facial muscle activity, head movements, shoulder shrugs). We report the findings of an individual with traumatic high-cervical spinal cord injury who coordinated reaching and grasping movements using his own paralysed arm and hand, reanimated through implanted FES, and commanded using his own cortical signals through an intracortical brain–computer interface (iBCI).
Methods: We recruited a participant into the BrainGate2 clinical trial, an ongoing study that obtains safety information regarding an intracortical neural interface device, and investigates the feasibility of people with tetraplegia controlling assistive devices using their cortical signals. Surgical procedures were performed at University Hospitals Cleveland Medical Center (Cleveland, OH, USA). Study procedures and data analyses were performed at Case Western Reserve University (Cleveland, OH, USA) and the US Department of Veterans Affairs, Louis Stokes Cleveland Veterans Affairs Medical Center (Cleveland, OH, USA). The study participant was a 53-year-old man with a spinal cord injury (cervical level 4, American Spinal Injury Association Impairment Scale category A). He received two intracortical microelectrode arrays in the hand area of his motor cortex, and 4 months and 9 months later received a total of 36 implanted percutaneous electrodes in his right upper and lower arm to electrically stimulate his hand, elbow, and shoulder muscles. The participant used a motorised mobile arm support for gravitational assistance and to provide humeral abduction and adduction under cortical control. We assessed the participant’s ability to cortically command his paralysed arm to perform simple single-joint arm and hand movements and functionally meaningful multi-joint movements. We compared iBCI control of his paralysed arm with that of a virtual three-dimensional arm. This study is registered with ClinicalTrials.gov, number NCT00912041.
Findings: The intracortical implant occurred on Dec 1, 2014, and we are continuing to study the participant. The last session included in this report was Nov 7, 2016. The point-to-point target acquisition sessions began on Oct 8, 2015 (311 days after implant). The participant successfully cortically commanded single-joint and coordinated multi-joint arm movements for point-to-point target acquisitions (80–100% accuracy), using first a virtual arm and second his own arm animated by FES. Using his paralysed arm, the participant volitionally performed self-paced reaches to drink a mug of coffee (successfully completing 11 of 12 attempts within a single session 463 days after implant) and feed himself (717 days after implant).
Interpretation: To our knowledge, this is the first report of a combined implanted FES+iBCI neuroprosthesis for restoring both reaching and grasping movements to people with chronic tetraplegia due to spinal cord injury, and represents a major advance, with a clear translational path, for clinically viable neuroprostheses for restoration of reaching and grasping after paralysis.
Funding: National Institutes of Health, Department of Veterans Affairs.