Researchers bypass the blood-brain barrier, widening treatment options for neurodegenerative and central nervous system disease
April 26, 2013
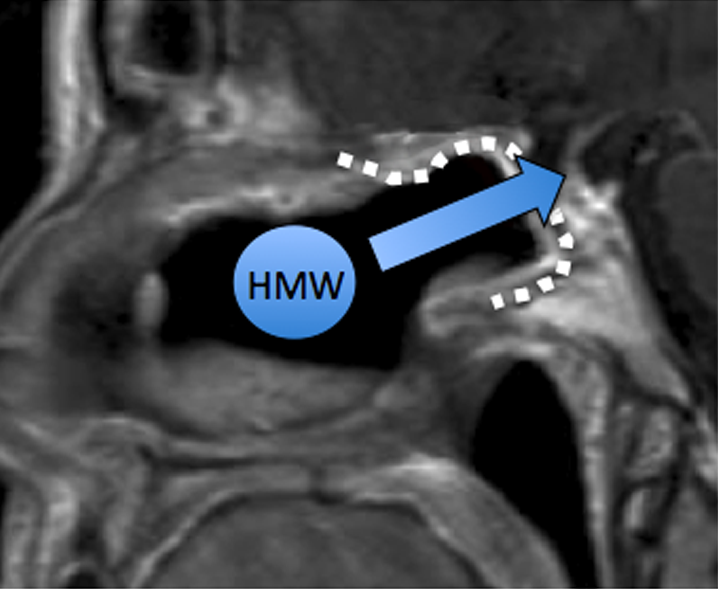
Sagittal MRI of a patient following endoscopic reconstruction of a skull base defect using a nasal mucosa graft (dotted white line; arrow denotes the proposed transmucosal pathway for HMW agents from the nose into the CNS through the graft) (credit: Benjamin S. Bleier et al./PLoS ONE)
The first known method to permanently bypass the blood-brain barrier*, using mucosa, or the lining of the nose, has been demonstrated by researchers in the department of Otology and Laryngology at the Massachusetts Eye and Ear/Harvard Medical School and the Biomedical Engineering Department of Boston University.
The method opens the door to new treatment options for those with neurodegenerative and CNS disease.
Inspired by recent advances in human endoscopic transnasal skull based surgical techniques, the investigators went to work to develop an animal model of this technique and use it to evaluate transmucosal permeability for the purpose of direct drug delivery to the brain.
In this study using a mouse model, researchers describe a novel method of creating a semi-permeable window in the blood-brain barrier using purely autologous (the patient’s own) tissues to allow for higher-molecular-weight drug delivery to the CNS. They demonstrated for the first time that these membranes are capable of delivering molecules to the brain that are up to 1,000 times larger than those excluded by the blood-brain barrier.
Neurodegenerative and central nervous system (CNS) diseases represent a major public health issue affecting at least 20 million children and adults in the United States alone.
Multiple drugs exist to treat and potentially cure these debilitating diseases, but 98 percent of all potential pharmaceutical agents are prevented from reaching the CNS directly due to the blood-brain barrier.
Many attempts have been made to deliver drugs across the blood-brain barrier using methods such as osmotic disruption and implantation of catheters into the brain; however, these methods are temporary and prone to infection and dislodgement.
“Since this is a proven surgical technique that is known to be safe and well tolerated, this data suggests that these membranes may represent the first known method to permanently bypass the blood-brain barrier using the patient’s own tissue,” said Benjamin S. Bleier, M.D., Otolaryngologist at Mass. Eye and Ear and HMS Assistant Professor. “This method may open the door for the development of a variety of new therapies for neurodegenerative and CNS disease.”
Study co-author Xue Han, Ph.D., an assistant professor of Biomedical Engineering at Boston University, said, “The development of this model enables us to perform critical preclinical testing of novel therapies for neurological and psychiatric diseases.”
Future studies will be directed towards developing clinical trials to test this method in patients who have already undergone these endoscopic surgeries.”
Their study is published on PLOS ONE (open access).
The study was supported by a grant from the Michael J. Fox Foundation for Parkinson’s Research and represents a collaborative effort between Mass. Eye and Ear and Dr. Xue Han of the Biomedical Engineering Department at Boston University.
* The blood–brain barrier separates circulating blood from the brain extracellular fluid (BECF) in the central nervous system (CNS).