Self-assembled nanoparticles release chemotherapy drug and heat to treat cancer
October 21, 2012
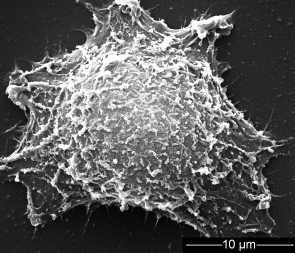
Gold nanorods deliver a chemotherapy drug to cancer cells and selectively release it when triggered by near-infrared light, along with heat, to zap cancer cells (credit: Xiao, Z. et al./Angewandte Chemie International Edition)
In new research from Brigham and Women’s Hospital (BWH), researchers have invented self-assembled, multifunctional, near-infrared-light-responsive nanoparticles to treat cancer.
The nanoparticles can deliver a chemotherapy drug specifically targeted to cancer cells and selectively release the drug in response to an external beam of light. They can also create heat for synergistic thermo-chemo-mediated anti-tumor effects.
Excitement around the potential for targeted nanoparticles (NPs) that can be controlled by a stimulus outside of the body for cancer therapy has been growing over the past few years. There has been considerable attention around near-infrared (NIR) light as an ideal method to stimulate nanoparticles from outside the body.
NIR is minimally absorbed by skin and tissue, it has the ability to penetrate deep tissue in a noninvasive way, and the energy from NIR light can be converted to heat by gold nanomaterials for effective thermal ablation of diseased tissue. This novel design takes the design a step further.
“The design of this gold nanorod and its self-assembly was inspired by nature and the ability of complementary strands of DNA to hybridize on their own without imposing complicated chemical processes on them,” explained Omid Farokhzad, MD, an anesthesiologist, director of the Laboratory of Nanomedicine and Biomaterials at BWH, and senior author of this study.
“Each functionalized DNA strand individually, and the self-assembled components as a system, play a distinct yet integrative role, resulting in synergistic targeted and triggered thermo-chemotherapy capable of eradicating tumors in our pre-clinical models.”
How the nanoparticles attack cancer cells
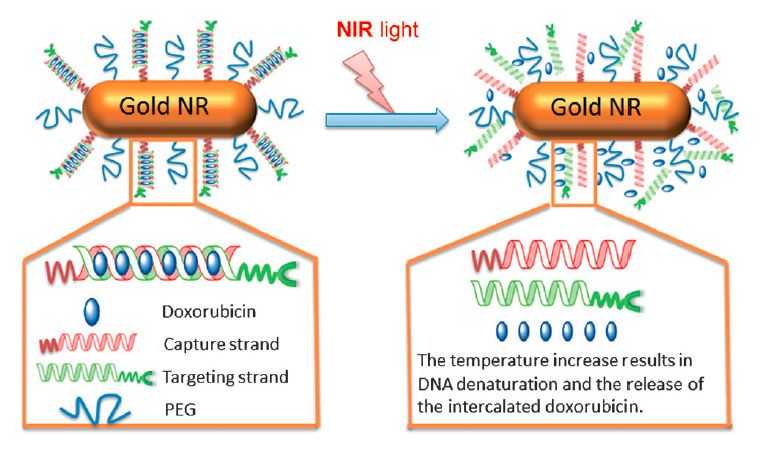
DNA assembly of a targeted, NIR-responsive delivery platform. This platform comprises gold NRs (50 nm x 10 nm nanorods), PEG layers, and complementary DNA oligonucleotides consisting of capture strands and targeting strands. Consecutive CG base pairs provide binding sites for doxorubicin (Dox) loading. The capture strands are conjugated to gold NRs for NIR response. The targeting strands are complementary to the capture strands and conjugated with ligands for molecular targeting. The delivery platform is assembled through the hybridization of capture strands attached on the NRs and targeting stands. The resulting double-stranded DNA structures form scaffolds for Dox intercalation. Upon NIR irradiation, the heated gold NRs result in DNA denaturation and the release of drugs (Dox) at the target site. (Credit: Xiao, Z. et al./Angewandte Chemie International Edition)
One DNA strand is attached to the gold nanorod and the complementary strand is attached to a stealth layer and a homing molecule that keeps the system “under the radar” of the immune system while targeting it directly to cancer cells. When the DNA strands come together, the targeted gold nanorod is formed and the double-stranded DNA serves as the scaffold for binding the chemotherapy drug, doxorubicin. The drug can be released in response to NIR light, which also results in generation of heat by the gold nanorods.
To demonstrate the robust capability of this nanorod system, Farokhzad and colleagues used a pre-clinical model to evaluate the in vivo anti-tumor efficacy in two different tumor models and four different groups with different drug regiments, each group varying in weight and tumor size.
Researchers injected the self-assembled nanoparticle and then 10 minutes post-injection, the tumors were irradiated using NIR light that activated the old nanorod to release the anti-cancer drugs and create heat. The results showed that this platform successfully eradicated tumors.
“Thermal ablation is already commonly used in cancer treatment,” said Dr. Farokhzad. “What is extremely exciting about this platform is that we are able to selectively target cancer cells and then hit the tumor twice: first with a controlled release of a chemotherapy drug and then secondly with triggered induction of heat from the activation of the gold nanorod. And all this can be done noninvasively.”
The researchers acknowledge that more research is necessary in other pre-clinical models before testing the safety and efficacy of this platform in human clinical trials.
This research was supported by the National Institutes of Health under grant number CA151884 and the David Koch-Prostate Cancer Foundation Program in Cancer Nanotherapeutics. O.C.F. has financial interest in BIND Biosciences, Selecta Biosciences, and Blend Therapeutics, biopharmaceutical companies that are developing therapeutic nanoparticles.