Single protein controls neurotransmitters
May 16, 2012
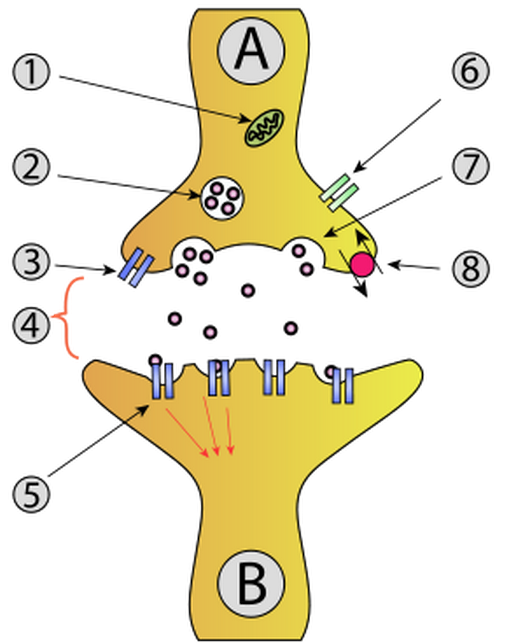
Increased expression of α2δ and β subunits leads to increased P/Q Ca2+ channel accumulation at synapses: (4) Synapse with neurotransmitter released; (6) Calcium channel (credit: Creative Commons)
Weill Cornell Medical College scientists have discovered that single protein alpha 2 delta controls the volume of neurotransmitters and other chemicals that flow between the synapses of brain neurons.
The study shows how brain cells talk to each other through these signals, relaying thoughts, feelings and action, and this powerful molecule plays a crucial role in regulating effective communication.
In the study, the investigators also suggest how the widely used pain drug Lyrica might work. The alpha 2 delta protein is the target of this drug and the new work suggests an approach to how other drugs could be developed that effectively turn particular neurotransmitter signals on and off to treat neurological disorders.
“We are amazed that any single protein has such power,” says the study’s lead investigator Dr. Timothy A. Ryan, professor of Biochemistry and associate professor of Biochemistry in Anesthesiology at Weill Cornell Medical College. “It is indeed rare to identify a biological molecule’s function that is so potent, that seems to be controlling the effectiveness of neurotransmission.”
The researchers found that alpha 2 delta determines how many calcium channels will be present at the synaptic junction between neurons. The transmission of chemical signals is triggered at the synapse by the entry of calcium into these channels, so the volume and speed of neurotransmission depends on the availability of these channels.
Researchers discovered that taking away alpha 2 delta from brain cells prevented calcium channels from getting to the synapse. “But if you add more alpha 2 delta, you can triple the number of channels at synapses,” Dr. Ryan says. “This change in abundance was tightly linked to how well synapses carry out their function, which is to release neurotransmitters.”
Before this study, it was known that Lyrica, which is used for neuropathic pain, seizures and fibromyalgia, binds to alpha 2 delta, but little was understood about how this protein works to control synapses.
Lifting up the Hood
Dr. Ryan is building what he calls a “shop manual” of neurological function, much of which centers on synaptic neurotransmission. In 2007 and 2008, he discovered crucial clues to how neurons repackage the chemicals used to signal across synapses. In 2011, Dr. Ryan discovered that distinct neurons differently tune the speed by which they package these chemicals. And in a recent study published April 29 in Nature Neuroscience, he described, for the first time, the molecular mechanisms at the synapse that control the release of dopamine, a crucial neurotransmitter.
“We are looking under the hood of these machines for the first time,” he says. “Many neurological diseases are considered to arise from pathologies of synaptic function. The synapse is so complex; at least a few thousand genes control how they work. Repairing them through treatment requires that we understand how they work.”
Dr. Ryan and his team often use two tools to conduct these studies — they pin fluorescent tags on to molecules involved in synaptic function, and use ultra-sensitive microscopy technology to watch these molecules up close and in real-time.
The researchers used the same toolkit to examine the function of calcium channels, which triggers neurotransmission. “At all synapses, the secretion of a neurotransmitter is driven by the arrival of an electric impulse, initiated by another neuron,” Dr. Ryan says. When this impulse arrives at the nerve terminal it triggers the opening of calcium channels. The calcium that rushes in is the key trigger that drives a synapse to secrete its neurotransmitter.
“We have known for the past half century that calcium is a key controller of neurotransmission,” he says. “Any small change in calcium influx has a big impact on neurotransmission.”
Protein Label
But the number of calcium channels at the synapse is not static. Neurons constantly replace worn out channels, and to do this, they build the channels in the neuron’s cell body and then package them up and ship them to the nerve terminal. In some cases, that is a very long journey — as much as a few feet, such as the distance between the brain and the base of the spinal cord or the length of a leg.
In the study, researchers tagged fluorescent proteins onto a gene that encodes protein that makes a calcium channel and delivered it to neurons. They then watched the progress of the newly formed channels as they made their way, from day four to day seven, from the bodies of neurons to the synapse.
They also manipulated the levels of alpha 2 delta, a suspected calcium channel partner, and discovered that when the protein was increased, more calcium channels were moved to the synapse. Less alpha 2 delta reduced the flow. “We discovered that alpha 2 delta made the decision of how many calcium channels should be shipped the length of the neuron to the synapse,” Dr. Ryan says. “It’s like the channels couldn’t be transported without an alpha 2 delta shipping label.”
The research team found however that alpha 2 delta must work in at least two steps. When they impaired a piece of alpha 2 delta that resembles proteins that are involved in how cells bind to each other, they found that this broken alpha 2 delta could still help get calcium channels shipped down to synapses. But once there, they no longer helped drive neurotransmitter release. “This means that not only does alpha 2 delta help to get calcium channels shipped out, but it also implies that something at the synapse has to sign-off on receiving the calcium channels, putting them in the right place for them to do their job,” Dr. Ryan says.
The researchers suggest that Lyrica might work by interfering with this final step since the piece of alpha 2 delta they “broke” that prevents the signing-off resembles parts of proteins that allows them to stick to each other in a kind of handshake.
These findings suggest that future therapies designed to manipulate neurotransmission could try to target this handshaking process, Dr. Ryan says. To do this will require that researchers identify the missing partner in the handshake.
“We hope these exciting findings are providing a new direction in how to make better drugs to control communication between brain cells,” Dr. Ryan says.
Ref.: Michael B. Hoppa, Beatrice Lana, Wojciech Margas, Annette C. Dolphin, Timothy A. Ryan, α2δ expression sets presynaptic calcium channel abundance and release probability, Nature, 2012, DOI: 10.1038/nature11033