Stroke damage in mice overcome by training that ‘rewires’ brain centers
February 7, 2013
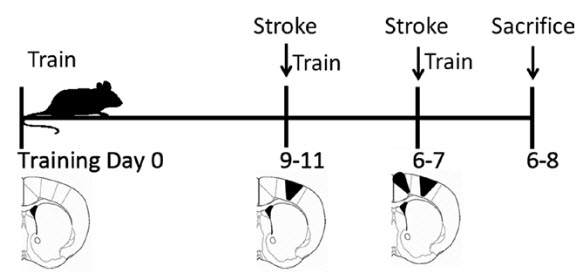
Schematic of time course for mouse familiarization, training, and stroke induction (credit: Steven R. Zeiler et al./Stroke)
Mice can recover from physically debilitating strokes that damage the primary motor cortex, the region of the brain that controls most movement in the body if the mice are quickly subjected to physical conditioning that rapidly “rewires” a different part of the brain to take over lost function, Johns Hopkins researchers have found. The research uses precise, intense and early treatment.
The researchers first trained normal but hungry mice to reach for and grab pellets of food in a precise way that avoided spilling the pellets and gave them the pellets as a reward. They reached maximum accuracy after seven to nine training days.
Then the researchers created experimental small strokes that left the mice with damage to the primary motor cortex. Predictably, the reaching and grasping precision disappeared, but a week of retraining, begun just 48 hours after the stroke, led the mice to again successfully perform the task with a degree of precision comparable to before the stroke.
Subsequent brain studies showed that although many nerve cells in the primary motor cortex were permanently damaged by the stroke, a different part of the brain called the medial premotor cortex adapted to control reaching and grasping.
“The function of the medial premotor cortex is not well-understood, but in this case it seemed to take over the functions associated with the reach-and-grab task in his experimental mice,” said study leader Steven R. Zeiler, M.D., Ph.D., an assistant professor of neurology at the Johns Hopkins University School of Medicine.
The researchers also report that otherwise healthy mice trained to reach and grasp pellets did not lose this ability after experiencing a stroke in the medial premotor cortex, which suggests that this part of the brain typically plays no role in those activities, and the existence of untapped levels of brain plasticity might be exploited to help human stroke victims.
Zeiler said another key finding in his research team’s mouse model was a reduction of the level of parvalbumin, a protein that marks the identity and activity of inhibitory neurons that keep the brain’s circuitry from overloading. With lower levels of parvalbumin in the medial premotor cortex, it appears the “brakes” are essentially off, allowing for the kind of activity required to reorganize and rewire the brain to take on new functions — in this case the ability to reach and grasp.
To prove that the learned functions had moved to the medial premotor cortex in the mice, the researchers induced strokes there. Again, the new skills were lost. And again, the mice could be retrained.
The research team’s next steps with their mouse model include evaluating the effect of drugs and timing of physical rehab on long-term recovery. The research could offer insight into whether humans should receive earlier and more aggressive rehab.
As many as 60 percent of stroke patients are currently left with diminished use of an arm or leg, and one-third need placement in a long-term care facility.
The research was supported by a startup fund from the Johns Hopkins University School of Medicine’s Department of Neurology as well as grants from the National Institutes of Health’s National Institute of Neurological Disorders and Stroke and the NIH’s National Institute of Mental Health.