Toward new drugs for the human and non-human cells in people
July 12, 2012
Almost 100 studies on a revolutionary approach to developing new medicines and treatments to target both the human and non-human components of people is reviewed in ACS’ Journal of Proteome Research.
Liping Zhao, Jeremy K. Nicholson and colleagues explain that human beings have been called “superorganisms” because their bodies contain 10 percent human cells and 90 percent microbes, which live mainly in the intestines.
Scientists thus are viewing people as vast ecosystems in which human, bacterial, fungal and other cells interact with each another. Microbes, for instance, release substances that determine whether human genes turn on or off and influence the immune system’s defenses against disease. And populations of microbes in the body change with changes in diet, medications and other factors.
“This superorganism view of the human body provides a complete new systems concept for managing human health at the clinically relevant whole body level,” say the authors. They term it “one of the most significant paradigm shifts in modern medicine.”
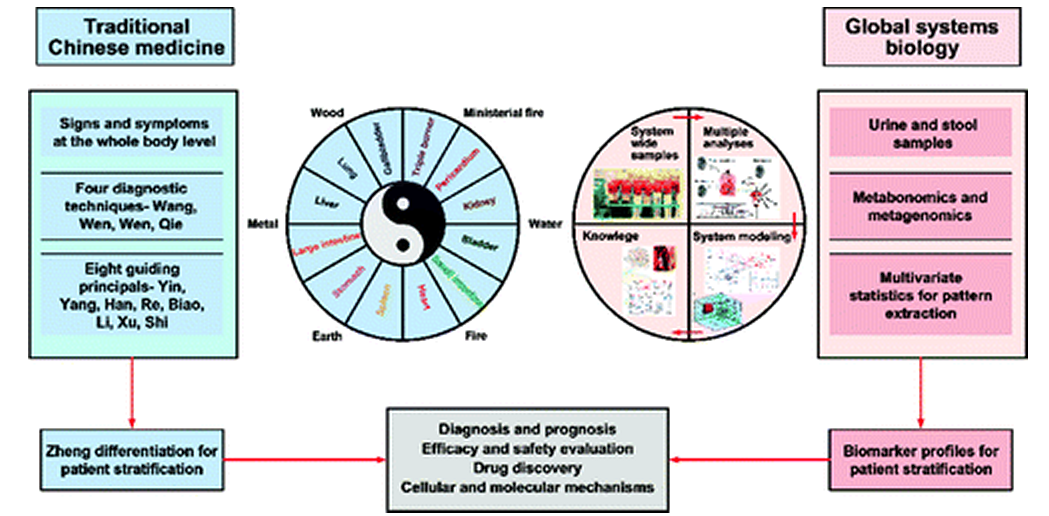
The article describes how this revolutionary change is fostering emergence of an approach called “functional metagenomics” for developing new medicines. It opens the possibility of sustaining health and treating disease with medicines and other substances that target non-human cells in the body. The article notes that many substances in traditional Chinese medicines may work in that way.
“There is widespread and increasing international interest in understanding the properties of traditional Chinese medicines (TCMs) for their potential utilization as a source of new drugs for Western markets,” the article notes. “Emerging evidence indicates that most TCM drugs are actually targeting both the host and its symbiotic microbes.”
The review “explores the challenges of and opportunities for harmonizing Eastern-Western drug discovery paradigms by focusing on emergent functions at the whole body level of humans as superorganisms. This could lead to new drug candidate compounds for chronic diseases targeting receptors outside the currently accepted “druggable genome” and shed light on current high interest issues in Western medicine such as drug-drug and drug-diet-gut microbial interactions that will be crucial in the development and delivery of future therapeutic regimes optimized for the individual patient.”
The authors acknowledge funding from the UK Foreign and Commonwealth Office/MOST and the Ministry of Science and Technology of the People’s Republic of China.
(Credit: ACS Journal of Proteome Research)