Wyss Institute to receive up to $37 million from DARPA to integrate multiple organ-on-chip systems to mimic the whole human body
July 25, 2012
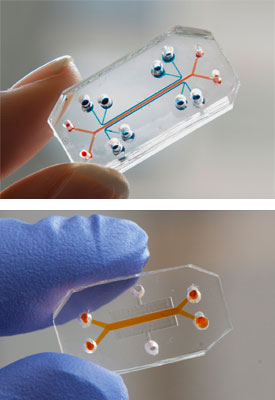
Wyss Institute researchers and a multidisciplinary team of collaborators seek to build and link 10 human organs-on-chips to mimic whole body physiology. The system will incorporate the Institute’s Human Lung-on-a-Chip (top) and Human Gut-on-a-Chip (bottom). (Credit: Wyss Institute)
The Wyss Institute for Biologically Inspired Engineering at Harvard University announced $37 million funding from the Defense Advanced Research Projects Agency (DARPA) to develop an automated instrument that integrates 10 human organs-on-chips to study complex human physiology outside the body.
This effort builds on the Institute’s past breakthroughs in which Institute researchers engineered microchips that recapitulate the microarchitecture and functions of living organs, such as the lung, heart, and intestine.
Each individual organ-on-chip is composed of a clear flexible polymer — about the size of a computer memory stick — that contains hollow microfluidic channels lined by living human cells.
Because the microdevices are translucent, they provide a window into the inner-workings of human organs without having to invade a living body.
With this new DARPA funding, Institute researchers and a multidisciplinary team of collaborators seek to build 10 different human organs-on-chips, to link them together to more closely mimic whole body physiology, and to engineer an automated instrument that will control fluid flow and cell viability while permitting real-time analysis of complex biochemical functions.
As an accurate alternative to traditional animal testing models that often fail to predict human responses, this instrumented “human-on-a-chip” will be used to rapidly assess responses to new drug candidates, providing critical information on their safety and efficacy.
Several U.S. agencies are working together to help safeguard Americans from deliberate chemical, biological, radiological, and nuclear threats, as well as from emerging infectious diseases, by drastically accelerating the drug development process.
As an example, DARPA, the National Institutes of Health (NIH), and the U.S. Food and Drug Administration (FDA) are actively collaborating to develop cutting edge technologies to predict drug safety.
This unique platform could help ensure that safe and effective therapeutics are identified sooner, and ineffective or toxic ones are rejected early in the development process. As a result, the quality and quantity of new drugs moving successfully through the pipeline and into the clinic may be increased, regulatory decision-making could be better informed, and patient outcomes could be improved.
Jesse Goodman, FDA Chief Scientist and Deputy Commissioner for Science and Public Health, commented that the automated human-on-chip instrument being developed “has the potential to be a better model for determining human adverse responses. FDA looks forward to working with the Wyss Institute in its development of this model that may ultimately be used in therapeutic development.”