Battery uses salinity difference between freshwater and saltwater
March 30, 2011
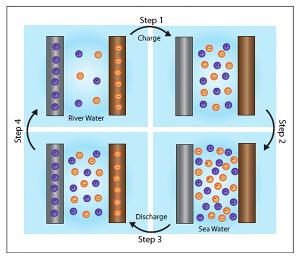
Purple and orange dots represent positively and negatively charged ions, respectively. In Step 1, a small electric current is applied to charge the battery. Step 2, the fresh water is purged and replaced with seawater. Step 3, electricity is drawn from the battery for use, draining the battery of its stored energy. Step 4, seawater is discharged and replaced with river water, for the cycle to begin anew. (Credit: Yi Cui, Stanford University)
Researchers at Stanford University have developed a battery that takes advantage of the difference in salinity between freshwater and seawater to produce electricity, says Yi Cui, associate professor of materials science and engineering, who led the research team.
The battery consists of two electrodes (one positive, one negative) immersed in an electrolyte with sodium and chlorine ions, the components of ordinary table salt.
The battery is filled with freshwater and a small electric current is applied to charge it up. The freshwater is then drained and replaced with seawater. Because seawater is salty, containing 60 to 100 times more ions than freshwater, it increases the voltage between the two electrodes.

The manganese dioxide nanorods used to make the postiive electrode of Yi Cui's new battery (credit: Yi Cui)
Cui’s team calculated that if all the world’s rivers were put to use, their batteries could supply about 2 terawatts of electricity annually, roughly 13 percent of the world’s current energy consumption.
A power plant operating with 50 cubic meters of freshwater per second could produce up to 100 megawatts of power, according to the team’s calculations. That would be enough to provide electricity for about 100,000 households, says Cui.
Ref.: Yi Cui et al., Batteries for Efficient Energy Extraction from a Water Salinity Difference, Nano Letters, March 17, 2011.