How the brain controls fat burning
January 16, 2015
(Credit: Monash University)
In case the “imaginary meal” approach to burning fat doesn’t work, now there’s a backup.
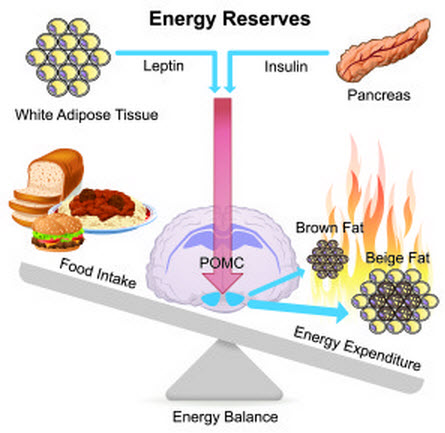
Monash University researchers have discovered that two naturally occurring hormones stimulate neurons in the brain’s hypothalamus, causing them to send signals through the nervous system that promote the conversion of white fat into brown fat. This leads to burning off excess fat.
The findings, published Thursday (Jan. 15) in the journal Cell, give new insights into how the brain regulates body fat, and may lead to more effective ways to lose weight and prevent obesity by promoting the conversion of white fat to brown fat.
This molecular mechanism depends on the combined action of leptin, an appetite suppressant generated in fat cells, and insulin, produced in the pancreas in response to rising levels of glucose in the blood.
How to burn fat
“These hormones give the brain a comprehensive picture of the fatness of the body,” said lead researcher Professor Tony Tiganis from the Department of Biochemistry and Molecular Biology. Leptin measures the level of existing fat reserves — the more fat, the more leptin — and insulin provides a measure of future fat reserves because glucose levels rise when we eat, Tiganis said.
Fat in adult humans is typically stored in adipocyte cells in white fat. But around the neck and shoulders, there is a second form of fat made of brown adipocytes. Rather than storing fat, these cells can be induced to burn it off.
The research team discovered leptin and insulin interact with proopiomelanocortin (POMC) neurons in the hypothalmus, which links the nervous system to the endocrine system via the pituitary gland.
In laboratory work, researchers were able to show that the process is regulated in these neurons by enzymes known as phosphatases, which inhibit the actions of each of the hormones. When the levels of these inhibitors were reduced, the browning and burning of fat increased.
Professor Tiganis said this fundamental process normally serves to maintain body weight, but in poor-diet-induced obesity this mechanism goes awry.
“Eventually, we think we may be able to help people lose weight by targeting these two enzymes. Turning white fat into brown fat is a very exciting new approach to developing weight loss agents. But it is not an easy task, and any potential therapy is a long way off,” Professor Tiganis said.
“One of the next big questions is whether the increased expression and prevention of insulin plus leptin signaling, and conversion of white to brown fat perturbs energy balance and promotes obesity,” said Tiganis. Another, said Garron Dodd, a postdoctoral fellow in Tiganis’s laboratory, is whether other parts of the brain are involved in signaling to and from adipose tissue.
Researchers from the Pennington Biomedical Research Center, Indiana University School of Medicine, Harvard Medical School, the University of Pennsylvania, and the University of Toronto collaborated on the research.
Abstract for Leptin and Insulin Act on POMC Neurons to Promote the Browning of White Fat
The primary task of white adipose tissue (WAT) is the storage of lipids. However, “beige” adipocytes also exist in WAT. Beige adipocytes burn fat and dissipate the energy as heat, but their abundance is diminished in obesity. Stimulating beige adipocyte development, or WAT browning, increases energy expenditure and holds potential for combating metabolic disease and obesity. Here, we report that insulin and leptin act together on hypothalamic neurons to promote WAT browning and weight loss. Deletion of the phosphatases PTP1B and TCPTP enhanced insulin and leptin signaling in proopiomelanocortin neurons and prevented diet-induced obesity by increasing WAT browning and energy expenditure. The coinfusion of insulin plus leptin into the CNS or the activation of proopiomelanocortin neurons also increased WAT browning and decreased adiposity. Our findings identify a homeostatic mechanism for coordinating the status of energy stores, as relayed by insulin and leptin, with the central control of WAT browning.