Lab-grown implanted neurons fuse with brain circuitry
November 25, 2011
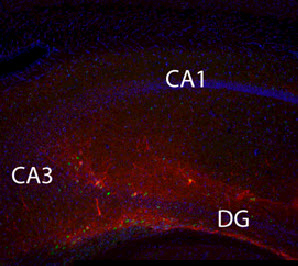
Fluorescent image of a mouse brain section in which transplanted hESC-derived neurons have been stained with human nuclear antigen and can be seen innervating various regions of the hippocampus (credit: Jason P. Weick et al./PNAS)
Neurons generated in the lab from blank-slate human embryonic stem cells (hESC) and implanted into the brains of mice can successfully fuse with the brain’s wiring and both send and receive signals, scientists st University of Wisconsin-Madison have found — a crucial step toward deploying customized cells to repair damaged or diseased brains.
“The big question was, can these cells integrate in a functional way?” says Jason P. Weick, the lead author of the new study and a staff scientist at the University of Wisconsin-Madison’s Waisman Center. “We show for the first time that these transplanted cells can both listen and talk to surrounding neurons of the adult brain.”
The Wisconsin team tested this by transplanting the human-derived neurons into the adult mouse hippocampus, an area of the brain that plays a key role in processing memory and spatial navigation. The capacity of the human cells to integrate into the mouse brain was observed in live tissue taken from the animals that received the cell transplants.
Weick and colleagues also reported that the human neurons adopted the rhythmic firing behavior of many brain cells talking to one another in unison. And, perhaps more importantly, that the human cells could modify the way the neural network behaved.
Specifically, the study demonstrated that hESC-derived neurons adopt the bursting behavior of a preexisting neural network, can modulate the mouse network activity via synaptic output, and can elicit spontaneous postsynaptic currents in hippocampal pyramidal neurons in slices taken from transplanted mouse brains. It also demonstrated that human neurons can make both excitatory and inhibitory synaptic connections with individual mouse neurons.
Optogenetics allows for precise, noninvasive stimulation
A critical tool that allowed the UW group to answer this question was optogenetics, where light instead of electric current is used to noninvasively stimulate only the transplanted human cells.
Weick explains that the capacity to modulate the implanted cells was a necessary step in determining the function of implanted cells, because previous technologies were too imprecise and unreliable to accurately determine what transplanted neurons were doing.
The appeal of human embryonic stem cells and induced pluripotent cells is the potential to manufacture limitless supplies of healthy, specialized cells to replace diseased or damaged cells. Brain disorders such as Parkinson’s disease and amyotrophic lateral sclerosis, more widely known as Lou Gehrig’s disease, are conditions that scientists think may be alleviated by using healthy lab grown cells to replace faulty ones. Multiple studies over the past decade have shown that both embryonic stem cells and induced cells can alleviate deficits of these disorders in animal models.
The new study opens the door to the potential for clinicians to deploy light-based stimulation technology to manipulate transplanted tissue and cells. “The marriage between stem cells and optogenetics has the potential to assist in the treatment of a number of debilitating neurodegenerative disorders,” notes Su-Chun Zhang, a UW-Madison professor of neuroscience. “You can imagine that if the transplanted cells don’t behave as they should, you could use this system to modulate them using light.”
Ref.: Jason P. Weick et al., Human embryonic stem cell-derived neurons adopt and regulate the activity of an established neural network, PNAS, 2011 [doi: 10.1073/pnas.1108487108]