Researchers restore leg movement in primates using wireless neural interface
November 10, 2016
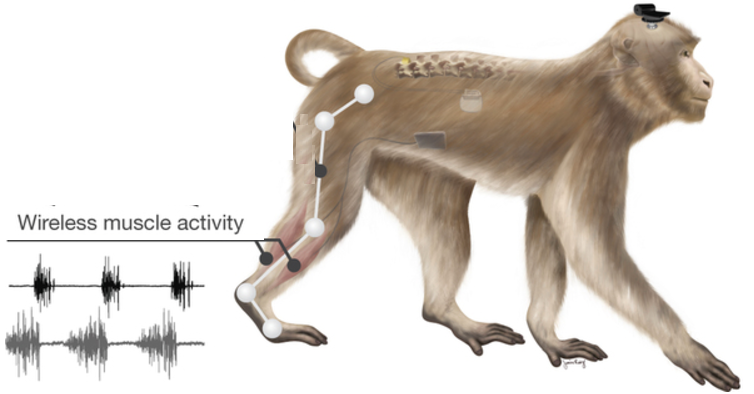
Brain-spinal interface bypasses spinal cord injuries in rhesus macaques, restoring nearly normal intentional walking movement (credit: Jemère Ruby)
An international team of scientists has used a wireless “brain-spinal interface” to bypass spinal cord injuries in a pair of rhesus macaques, restoring nearly normal intentional walking movement to a temporarily paralyzed leg.
The finding could help in developing a similar system to rehabilitate humans who have had spinal cord injuries.
The system uses signals recorded from a pill-sized electrode array implanted in the motor cortex of the brain to trigger coordinated electrical stimulation of nerves in the spine that are responsible for locomotion.
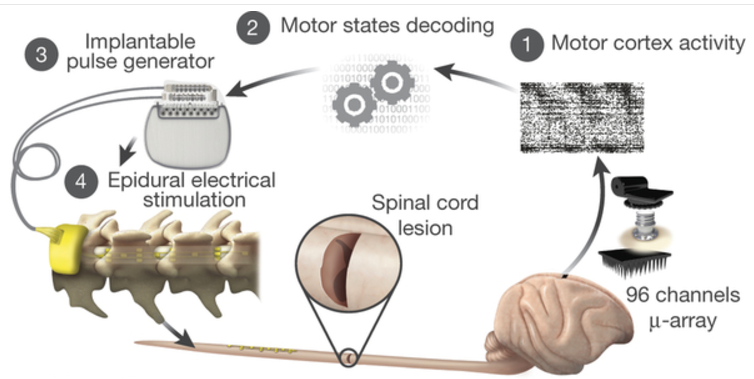
Monkeys were implanted with a microelectrode array into the leg area of the left motor cortex. (1) During recordings, a wireless module transmitted broadband neural signals to a control computer. A raster plot recorded three successive gait cycles. Each line represents spiking events identified from one electrode, while the horizontal axis indicates time. (2) A decoder running on the control computer identified motor states from these neural signals. (3) These motor states triggered electrical spinal cord stimulation protocols, using an implanted pulse generator with real-time triggering capabilities. (4) The stimulator was connected to a spinal implant targeting specific dorsal roots of the lumbar spinal cord. Electromyographic (muscle) signals of an extensor (gray) and flexor (black) muscles acting at the ankle recorded over three successive gait cycles are shown together with a stick diagram decomposition of leg movements during the stance (gray) and swing (black) phases of gait. (credit: Jemère Ruby)
A wireless neurosensor, developed in the neuroengineering lab of Brown University professor Arto Nurmikko, sends the signals gathered by the brain chip wirelessly to a computer that decodes them and sends them wirelessly back to an electrical spinal stimulator implanted in the lumbar spine, below the area of injury.
That electrical stimulation, delivered in patterns coordinated by the decoded brain, sends signals to the spinal nerves that control locomotion.

The sensor technology was developed in part for investigational use in humans by the BrainGate collaboration, a research team that includes Brown, Case Western Reserve University, Massachusetts General Hospital, the Providence VA Medical Center, and Stanford University. The technology is being used in ongoing pilot clinical trials, and was used previously in a study in which people with tetraplegia were able to operate a robotic arm simply by thinking about the movement of their own hand. (credit: Brown University)
The ability to transmit brain signals wirelessly was critical to this work with monkeys, the researchers note. Wired brain-sensing systems limit freedom of movement, which in turn limits the information researchers are able to gather about locomotion.
Despite current limitations, the research sets the stage for future studies in primates and, at some point, potentially as a rehabilitation aid in humans, the researchers suggest.
The study, published in the journal Nature, was performed by scientists and neuroengineers in a collaboration led by Ecole Polytechnique Federale Lausanne (EPFL) in Switzerland, together with Brown University, Medtronic and Fraunhofer ICT-IMM in Germany.
The research was funded by European Community’s Seventh Framework Program, International Foundation for Research in Paraplegia Starting Grant from the European Research Council, The Wyss Centre in Geneva Marie Curie Fellowship, Marie Curie COFUND EPFL fellowships, Medtronic Morton Cure Paralysis Fund fellowship, NanoTera.ch Programme (SpineRepair), National Centre of Competence in Research in Robotics Sinergia program, Sino-Swiss Science and Technology Cooperation, and the Swiss National Science Foundation.
Abstract of A brain–spine interface alleviating gait deficits after spinal cord injury in primates
Spinal cord injury disrupts the communication between the brain and the spinal circuits that orchestrate movement. To bypass the lesion, brain–computer interfaces have directly linked cortical activity to electrical stimulation of muscles, and have thus restored grasping abilities after hand paralysis. Theoretically, this strategy could also restore control over leg muscle activity for walking. However, replicating the complex sequence of individual muscle activation patterns underlying natural and adaptive locomotor movements poses formidable conceptual and technological challenges. Recently, it was shown in rats that epidural electrical stimulation of the lumbar spinal cord can reproduce the natural activation of synergistic muscle groups producing locomotion. Here we interface leg motor cortex activity with epidural electrical stimulation protocols to establish a brain–spine interface that alleviated gait deficits after a spinal cord injury in non-human primates. Rhesus monkeys (Macaca mulatta) were implanted with an intracortical microelectrode array in the leg area of the motor cortex and with a spinal cord stimulation system composed of a spatially selective epidural implant and a pulse generator with real-time triggering capabilities. We designed and implemented wireless control systems that linked online neural decoding of extension and flexion motor states with stimulation protocols promoting these movements. These systems allowed the monkeys to behave freely without any restrictions or constraining tethered electronics. After validation of the brain–spine interface in intact (uninjured) monkeys, we performed a unilateral corticospinal tract lesion at the thoracic level. As early as six days post-injury and without prior training of the monkeys, the brain–spine interface restored weight-bearing locomotion of the paralysed leg on a treadmill and overground. The implantable components integrated in the brain–spine interface have all been approved for investigational applications in similar human research, suggesting a practical translational pathway for proof-of-concept studies in people with spinal cord injury.