A cheap, long-lasting, sustainable battery for grid energy storage
September 2, 2016
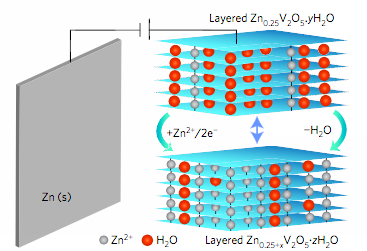
The zinc-ion battery consists of a water-based electrolyte, a pillared vanadium oxide positive electrode (right), and an inexpensive metallic zinc negative electrode (left). The battery generates electricity through a reversible process called intercalation, where positively-charged zinc ions are oxidized from the zinc metal negative electrode, travel through the electrolyte and insert between the layers of vanadium oxide nanosheets in the positive electrode. This drives the flow of electrons in the external circuit, creating an electrical current. The reverse process occurs on charge. (credit: Dipan Kundu et al./Nature Energy)
University of Waterloo chemists have developed a long-lasting, safe, zinc-ion battery that costs half the price of current lithium-ion batteries. It could help communities shift from traditional power plants to renewable solar and wind energy production, where electricity storage overnight is needed.
The battery is water-based and uses cheap but safe, non-flammable, non-toxic materials, compared to expensive, flammable, organic electrolytes in lithium-ion batteries, which are used in the exploding Samsung Galaxy Note 7 smartphones reported last week and in previously reported exploding hoverboards.
Where cost, safety, and life cycle are vital, not size
Lithium-ion batteries have much higher energy density (energy that can be stored per unit volume) than water-based batteries (making lithium-ion batteries attractive for smartphones and other compact devices), but water-based zinc-ion batteries are more feasible for grid-scale applications, where cost, safety, and life cycle are important, not size.
The cell design satisfies four vital criteria: high reversibility, rate, capacity, and no zinc dendrite formation. It provides more than 1,000 cycles. Lithium-ion batteries also operate by intercalation (of lithium ions) but they typically use expensive, flammable, organic electrolytes.
The bonus for manufacturers is they can produce this zinc battery at low cost because its fabrication does not require special conditions, such as ultra-low humidity or the handling of flammable materials needed for lithium ion batteries, the chemists say.
“The focus used to be on minimizing size and weight for the portable electronics market and cars,” said Dipan Kundu, a University of Waterloo postdoctoral fellow and the paper’s first author. “Grid storage needs a different kind of battery and that’s given us license to look into different materials.”
The discovery appears in the journal Nature Energy.
Abstract of A high-capacity and long-life aqueous rechargeable zinc battery using a metal oxide intercalation cathode
Although non-aqueous Li-ion batteries possess significantly higher energy density than their aqueous counterparts, the latter can be more feasible for grid-scale applications when cost, safety and cycle life are taken into consideration. Moreover, aqueous Zn-ion batteries have an energy storage advantage over alkali-based batteries as they can employ Zn metal as the negative electrode, dramatically increasing energy density. However, their development is plagued by a limited choice of positive electrodes, which often show poor rate capability and inadequate cycle life. Here we report a vanadium oxide bronze pillared by interlayer Zn2+ ions and water (Zn0.25V2O5⋅nH2O), as the positive electrode for a Zn cell. A reversible Zn2+ ion (de)intercalation storage process at fast rates, with more than one Zn2+ per formula unit (a capacity up to 300 mAh g−1), is characterized. The Zn cell offers an energy density of ∼450 Wh l−1 and exhibits a capacity retention of more than 80% over 1,000 cycles, with no dendrite formation at the Zn electrode.