Attacking Alzheimer’s with ultrasound
February 20, 2015
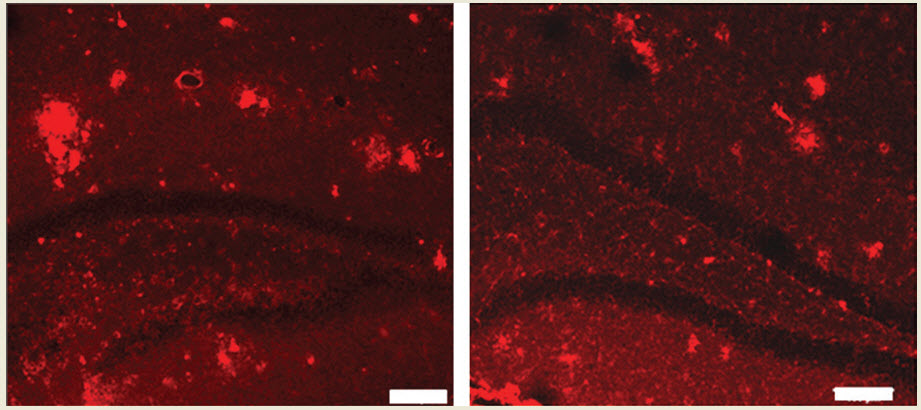
The plaque abnormalities on untreated transgenic mice (left) and the brain of a transgenic mouse that has been treated with MR imaging-guided focused ultrasound (right), showing reduced plaque (credit: Kullervo Hynynen, Sunnybrook Research Institute)
For the first time, researchers have reversed some of the symptoms of Alzheimer’s disease* in mice using magnetic resonance (MR) imaging-guided focused ultrasound.
As KurzweilAI reported in 2012, Sunnybrook Research Institute scientists used MR imaging-guided focused ultrasound to temporarily open the blood-brain barrier (BBB), allowing for more effective delivery of drugs to the brain. The method uses a microbubble contrast agent. The microbubbles vibrate when they pass through the ultrasound beam, temporarily creating an opening in the BBB for the drugs to pass through. In addition, this combination of ultrasound and microbubbles has been shown to increase the number of new neurons and the dendrite length.
In the new study, Kullervo Hynynen, Ph.D., a medical physicist at Sunnybrook Research Institute, and his collaborators studied the effects of using MR imaging-guided focused ultrasound on the hippocampus of transgenic (TgCRND8) mice.
Mice with this genetic variant have increased plaque on their hippocampus, the part of the brain that helps convert information from short-term to long-term memory; they also display symptoms similar to Alzheimer’s such as memory impairment and learning reversal. That allows these transgenic mice to be used as an animal model for Alzheimer’s disease.
Improved cognition and spatial learning
The researchers used MR imaging-guided focused ultrasound with microbubbles to open the BBB and treat the hippocampus of the mice. The hippocampus is divided into two parts, one in each hemisphere of the brain. They found the treatment led to improvements in cognition and spatial learning in the transgenic mice, potentially caused by reduced plaque and increased neuronal plasticity due to the focused ultrasound treatment.
They found no tissue damage or negative behavioral changes in the mice due to the treatments in either the transgenic mice or the control (nontransgenic) mice. Both groups of mice benefited from increased neuronal plasticity, which confirms the previous research on the effects of MR imaging-guided focused ultrasound on plasticity in healthy mice.
How it works
According to the Radiology paper, the investigators in previous studies have suggested two potential mechanisms for plaque reduction:
- Opening of the BBB permits the entry of endogenous immunoglobuline G and immunoglobulin M from the periphery into the brain, which assists with plaque clearance.
- MR imaging-guided focused ultrasound causes mild activation of astrocytes and microglia, which were shown to internalize amyloid and contribute to plaque reduction. These potential mechanisms are likely to also contribute to the reduced plaque observed in this study.
Next steps
“The results are an exciting step in the search for Alzheimer’s treatments,” said Steven Krosnick, M.D., Program Director for Image-Guided Interventions at the National Institute of Biomedical Imaging and Bioengineering at NIH, “but there is more to be done. There are limitations on the memory tests that can be done on mice, and human cognition is significantly more complex.
“Hopefully these results will open doors to more research on how MR imaging-guided focused ultrasound could benefit cognition and perhaps be magnified by using other therapeutics in conjunction with this method.”
This research was supported in part by the National Institute of Biomedical Imaging and Bioengineering award #EB003268
* An estimated 5.2 million Americans suffer from Alzheimer’s. It is the sixth leading cause of death in the United States and there is currently no treatment for the disease.
Abstract for Alzheimer disease in a mouse model: MR imaging–guided focused ultrasound targeted to the hippocampus opens the blood-brain barrier and improves pathologic abnormalities and behavior
Purpose: To validate whether repeated magnetic resonance (MR) imaging–guided focused ultrasound treatments targeted to the hippocampus, a brain structure relevant for Alzheimer disease (AD), could modulate pathologic abnormalities, plasticity, and behavior in a mouse model.
Materials and Methods: All animal procedures were approved by the Animal Care Committee and are in accordance with the Canadian Council on Animal Care. Seven-month-old transgenic (TgCRND8) (Tg) mice and their nontransgenic (non-Tg) littermates were entered in the study. Mice were treated weekly with MR imaging–guided focused ultrasound in the bilateral hippocampus (1.68 MHz, 10-msec bursts, 1-Hz burst repetition frequency, 120-second total duration). After 1 month, spatial memory was tested in the Y maze with the novel arm prior to sacrifice and immunohistochemical analysis. The data were compared by using unpaired t tests and analysis of variance with Tukey post hoc analysis.
Results: Untreated Tg mice spent 61% less time than untreated non-Tg mice exploring the novel arm of the Y maze because of spatial memory impairments (P < .05). Following MR imaging–guided focused ultrasound, Tg mice spent 99% more time exploring the novel arm, performing as well as their non-Tg littermates. Changes in behavior were correlated with a reduction of the number and size of amyloid plaques in the MR imaging–guided focused ultrasound– treated animals (P < .01). Further, after MR imaging–guided focused ultrasound treatment, there was a 250% increase in the number of newborn neurons in the hippocampus (P < .01). The newborn neurons had longer dendrites and more arborization after MR imaging– guided focused ultrasound, as well (P < .01).
Conclusion: Repeated MR imaging–guided focused ultrasound treatments led to spatial memory improvement in a Tg mouse model of AD. The behavior changes may be mediated by decreased amyloid pathologic abnormalities and increased neuronal plasticity.