Controlling brain cells with ultrasound
September 15, 2015

For the first time, sound waves are used to control brain cells. Salk scientists developed the new technique, dubbed sonogenetics, to selectively and noninvasively turn on groups of neurons in worms that could be a boon to science and medicine. (credit: Salk Institute)
Salk scientists have developed a new method, dubbed sonogenetics, to selectively activate brain, heart, muscle and other cells using ultrasonic waves (the same type of waves used in medical sonograms).
This new method may have advantages over the similar light-based approach known as optogenetics, particularly for human therapeutics. It is described today (Sept. 15, 2015) in the journal Nature Communications.
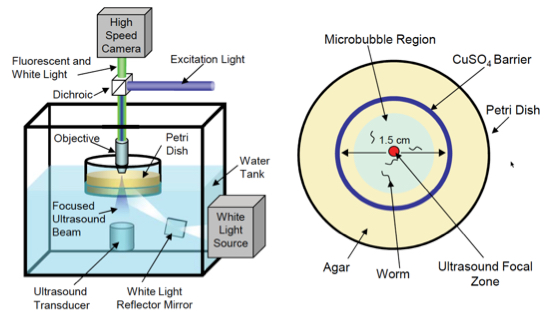
Experiment setup. (Left) Schematic of the computer-controlled imaging and ultrasound exposure system. (Right) Top-down view of petri dish with agar (food) plate with animals corralled into a small area by a copper barrier. (credit: Stuart Ibsen et al./Nature Communications)
Sreekanth Chalasani, an assistant professor in Salk’s Molecular Neurobiology Laboratory and senior author of the study, and his colleagues activated neurons in the nematode C. elegans that don’t usually react to ultrasound.
They found that microbubbles of gas outside of the worm were able to amplify the low-intensity ultrasound waves. “The microbubbles grow and shrink in [sync] with the ultrasound pressure waves,” Ibsen says. “These oscillations can then propagate noninvasively into the worm.”
They also found that when the ultrasound hits gas bubbles, they cause a membrane ion channel, TRP-4, to open up and activate the cell. Armed with that knowledge, the team tried adding the TRP-4 channel to neurons that don’t normally have it.
Possible human uses next
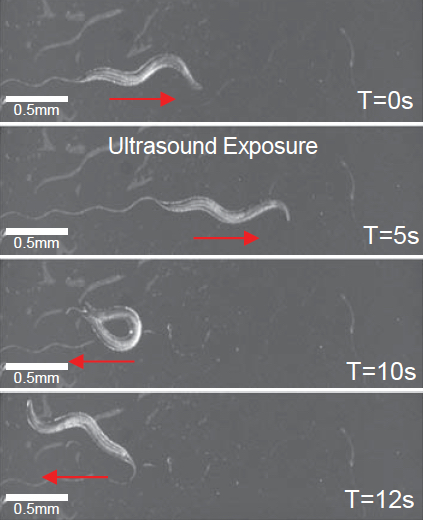
Worm exhibits reversals and omega bends upon ultrasound stimulus (single 10 ms pulse at 2.25 MHz with peak negative pressure of 0.9 MPa) in the presence of micro bubbles (credit: Stuart Ibsen et al./Nature Communications)
So far, sonogenetics has only been applied to C. elegans neurons. But TRP-4 could be added to any calcium-sensitive cell type in any organism including humans, Chalasani says. Here’s how it would work:
- The microbubbles are injected into the bloodstream, and are distributed throughout the body —- an approach already used in some human imaging techniques.
- Ultrasound could then noninvasively reach any tissue of interest, including the brain, be amplified by the microbubbles, and activate the cells of interest through TRP-4. (Many cells in the human body, he points out, can respond to the influxes of calcium caused by TRP-4.)
The researchers have already begun testing the approach in mice.
Both optogenetics and sonogenetics approaches, Chalasani says, hold promise in basic research by letting scientists study the effect of cell activation. And they also may be useful in therapeutics through the activation of cells affected by disease.
However, for either technique to be used in humans, researchers first need to develop safe ways to deliver the light or ultrasound-sensitive channels to target cells.
The work and the researchers involved were supported by a Salk Institute Pioneer Fund Postdoctoral Fellowship, a Salk Institute Innovation Grant, the Rita Allen Foundation, the W.M. Keck Foundation and the National Institutes of Health. A University of California, San Diego researcher was also involved in the research.
Abstract of Sonogenetics is a non-invasive approach to activating neurons in Caenorhabditis elegans
A major challenge in neuroscience is to reliably activate individual neurons, particularly those in deeper brain regions. Current optogenetic approaches require invasive surgical procedures to deliver light of specific wavelengths to target cells in order to activate or silence them. Here, we demonstrate the use of low-pressure ultrasound as a non-invasive trigger to activate specific ultrasonically-sensitized neurons in the nematode, Caenorhabditis elegans. We first show that wild-type animals are insensitive to low pressure ultrasound and require gas-filled microbubbles to transduce the ultrasound wave. We find that neuron-specific misexpression of TRP-4, the pore-forming subunit of a mechanotransduction channel, sensitizes neurons to ultrasound stimulus resulting in motor outputs. Furthermore, we use this approach to manipulate the function of sensory neurons and interneurons and identify a role for the PVD sensory neurons in modifying locomotory behaviors. We suggest this method can be broadly applied to manipulate cellular functions in vivo.