Forcing cancer cells to devour themselves by blocking a protein signal
April 9, 2014
Cancer cell death by autophagy (Credit: Cell Reports)
Under stress from chemotherapy or radiation, some cancer cells dodge death by autophagy — eating a bit of themselves — allowing them to essentially sleep through treatment and later awaken as tougher, resistant disease.
But interfering with a single cancer-promoting protein and its receptor can turn this resistance mechanism into lethal, runaway self-cannibalization, researchers at The University of Texas MD Anderson Cancer Center report in the journal Cell Reports (open access).
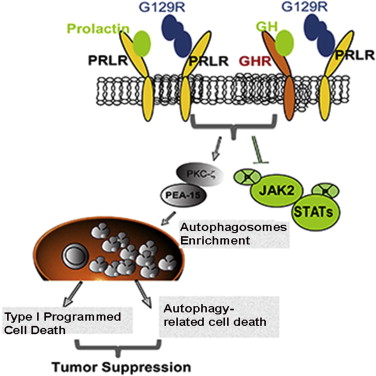
“When we block prolactin signaling, it sets off a chain of downstream events that result in cell death by autophagy,” said senior author Anil Sood, M.D., professor of Gynecologic Oncology and Reproductive Medicine. “Prolactin is a potent growth factor for many types of cancers, including ovarian cancer.”
Autophagy — self-eating — is a natural cellular defense against lack of nutrients and other stressors. It also recycles damaged organelles and proteins for new use by the cell. Autophagy puts the cell in an inactive quiet state called quiescence, which allows it to recover, Sood said. For cancer cells, it’s a way to survive treatment.
“Our findings provide a clinical rationale for blocking prolactin and its receptor and for using prolonged autophagy as an alternative strategy for treating cancers,” said Yunfei Wen, Ph.D., first author of this study and instructor of Gynecologic Oncology.
Steep reductions in tumor weight for mice with ovarian cancer
Prolactin (PRL) is a hormone previously implicated in ovarian, endometrial and other cancer development and progression. When PRL binds to its cell membrane receptor, PRLR, activation of cancer-promoting cell signaling pathways follows. Poor understanding of the underlying processes has made the pathway hard to target for cancer treatment, Sood said.
Given that knowledge, the researchers started with mouse experiments and worked backward to cell line experiments, a reversal of the usual order of preclinical cancer research.
A slight variant of normal prolactin called G129R interferes with the connection between prolactin and its receptor. Using G129R, Sood and colleagues treated mice that had two different lines of human ovarian cancer that each expresses the prolactin receptor.
After 28 days of treatment with G129R, tumor weights fell by 50 percent for mice with either type of ovarian cancer. Adding the taxane-based chemotherapy agent paclitaxel, commonly used to treat ovarian cancer, cut tumor weight by 90 percent. Higher doses of G129R may result in even greater therapeutic benefit, Sood said.
The mice did not otherwise lose weight, suffer lowered blood counts or show any other sign of toxicity of side effects from G129R treatment in the liver, spleen or kidneys.
3D experiments show death by self-eating
The team used three-dimensional culture of cancer spheroids, where treatment with the prolactin-mimicking peptide sharply reduced the number of spheroids. Treatment also blocked the activation of JAK2 and STAT signaling pathways known to promote cancer.
Protein analysis in the treated spheroids showed increased presence of autophagy factors and genomic analysis revealed increased expression of a number of genes involved in autophagy progression and cell death.
A series of experiments using fluorescence and electron microscopy showed that the cytosol of treated cells had large numbers of cavities caused by autophagy, a hallmark of autophagy-induced cell death.
Autophagy works by encasing targeted proteins or organelles in a membrane, which then connects with lysosomes that dissolve the contents, leaving empty cavities, or vacuoles. Adding an autophagy inhibitor reversed the treatment effect of G129R in the 3D spheres.
Connection to ovarian cancer patient survival
The team also connected the G129R-induced autophagy to the activity of PEA-15, a known cancer inhibitor. Analysis of tumor samples from 32 ovarian cancer patients showed that tumors express higher levels of the prolactin receptor and lower levels of phosphorylated PEA-15 than normal ovarian tissue.
Patients with low levels of the prolactin receptor and higher PEA-15 had longer overall survival than those with high PRLR and low PEA-15.
The researchers are working to take G129R to clinical trials said Sood, who holds the Bettyann Asche Murray Distinguished Professorship in Ovarian Cancer Research.
Abstract of Cell Reports paper
- PRLR antagonism by G129R inhibits growth of ovarian cancer or cancer spheroids
- Autophagy-related cell death is induced by G129R in cancer spheroids
- Pea-15 functions as a tumor suppressor to mediate G129R-induced autophagy
- Tumoral PRL/PRLR correlates inversely with autophagic factors in clinical samples
Therapeutic upregulation of macroautophagy in cancer cells provides an alternative mechanism for cell death. Prolactin (PRL) and its receptor (PRLR) are considered attractive therapeutic targets because of their roles as growth factors in tumor growth and progression. We utilized G129R, an antagonist peptide of PRL, to block activity of the tumoral PRL/PRLR axis, which resulted in inhibition of tumor growth in orthotopic models of human ovarian cancer. Prolonged treatment with G129R induced the accumulation of redundant autolysosomes in 3D cancer spheroids, leading to a type II programmed cell death. This inducible autophagy was a noncanonical beclin-1-independent pathway and was sustained by an astrocytic phosphoprotein (PEA-15) and protein kinase C zeta interactome. Lower levels of tumoral PRL/PRLR in clinical samples were associated with longer patient survival. Our findings provide an understanding of the mechanisms of tumor growth inhibition through targeting PRL/PRLR and may have clinical implications.