Functional thymus organ grown in mice from lab-created cells
August 26, 2014
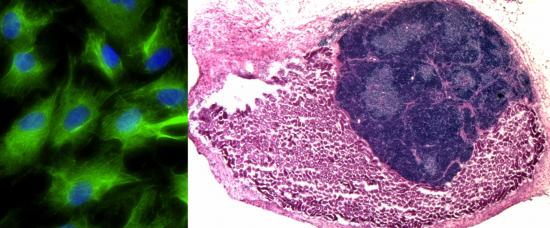
Fibroblasts transformed into induced thymic epithelial cells (iTEC) in vitro (left, iTEC shown in green). iTEC transplanted onto the mouse kidney forms an organized and functional mini-thymus (right, kidney cells in pink, thymus cells in dark blue) (credit: MRC)
Scientists have for the first time grown a complex, fully functional organ from scratch in a living animal by transplanting cells that were originally created in a laboratory to form a replacement thymus*, a vital organ of the immune system.
The advance could in the future aid the development of “lab-grown” replacement organs.
Researchers from the MRC Centre for Regenerative Medicine, at the University of Edinburgh, took cells called fibroblasts from a mouse embryo and converted them directly into a completely unrelated type of cell — specialized thymus cells — using a technique called “reprogramming.”
When mixed with other thymus cell types and transplanted into mice, these cells formed a replacement organ that had the same structure, complexity and function as a healthy native adult thymus. The reprogrammed cells were also capable of producing T cells in the lab, a type of white blood cell important for fighting infection.
Medical Research Council | Scientists have for the first time grown a complex, fully functional organ from scratch in a living animal by transplanting cells that were originally created in a laboratory.
Thymus transplant treatment
The researchers hope that with further refinement their lab-made cells could form the basis of a readily available thymus transplant treatment for people with a weakened immune system. They may also enable the production of patient-matched T cells. The research is published in the journal Nature Cell Biology.
Being able to create a complete transplantable thymus from cells in a lab would be a huge step forward in treating such conditions. And while several studies have shown it is possible to produce collections of distinct cell types in a dish, such as heart or liver cells, scientists haven’t yet been able to grow a fully intact organ from cells created outside the body.
The researchers carried out their study using fibroblast cells taken from mouse embryos. By increasing levels of a protein called FOXN1, which guides development of the thymus during normal organ development in the embryo, they were able to directly reprogram these cells to become a type of thymus cell called thymic epithelial cells. These are the cells that provide the specialist functions of the thymus, enabling it to make T cells.
The induced thymic epithelial cells (iTEC) were then combined with other thymus cells (to support their development) and grafted onto the kidneys of genetically identical mice. After four weeks, the cells had produced well-formed organs with the same structure as a healthy thymus, with clearly defined regions (known as the cortex and medulla). The iTEC cells were also able to produce different types of T cells from immature blood cells in the lab.
The study was funded by Leukaemia & Lymphoma Research, Darwin Trust of Edinburgh, the MRC and the European Union Seventh Framework Program.
* The thymus, located near the heart, is a vital organ of the immune system. It produces T cells, which guard against disease by scanning the body for malfunctioning cells and infections. When they detect a problem, they mount a coordinated immune response that tries to eliminate harmful cells, such as cancer, or pathogens like bacteria and viruses.
People without a fully functioning thymus can’t make enough T cells and as a result are very vulnerable to infections. This can be a particular problem for patients who need a bone marrow transplant (for example to treat leukemia), since a functioning thymus is needed to rebuild the immune system once the transplant has been received. The problem can also affect children; around one in 4,000 babies born each year in the UK have a malfunctioning or completely absent thymus (due to conditions such as DiGeorge syndrome).
Thymus disorders can sometimes be treated with infusions of extra immune cells, or transplantation of a thymus organ soon after birth, but both are limited by a lack of donors and problems matching tissue to the recipient.
Abstract of Nature Cell Biology paper
A central goal of regenerative medicine is to generate transplantable organs from cells derived or expanded in vitro. Although numerous studies have demonstrated the production of defined cell types in vitro, the creation of a fully intact organ has not been reported. The transcription factor forkhead box N1 (FOXN1) is critically required for development of thymic epithelial cells (TECs), a key cell type of the thymic stroma. Here, we show that enforced Foxn1 expression is sufficient to reprogramme fibroblasts into functional TECs, an unrelated cell type across a germ-layer boundary. These FOXN1-induced TECs (iTECs) supported efficient development of both CD4+ and CD8+ T cells in vitro. On transplantation, iTECs established a complete, fully organized and functional thymus, that contained all of the TEC subtypes required to support T-cell differentiation and populated the recipient immune system with T cells. iTECs thus demonstrate that cellular reprogramming approaches can be used to generate an entire organ, and open the possibility of widespread use of thymus transplantation to boost immune function in patients.