How a synapse stays stable when its proteins are renewed
April 30, 2014
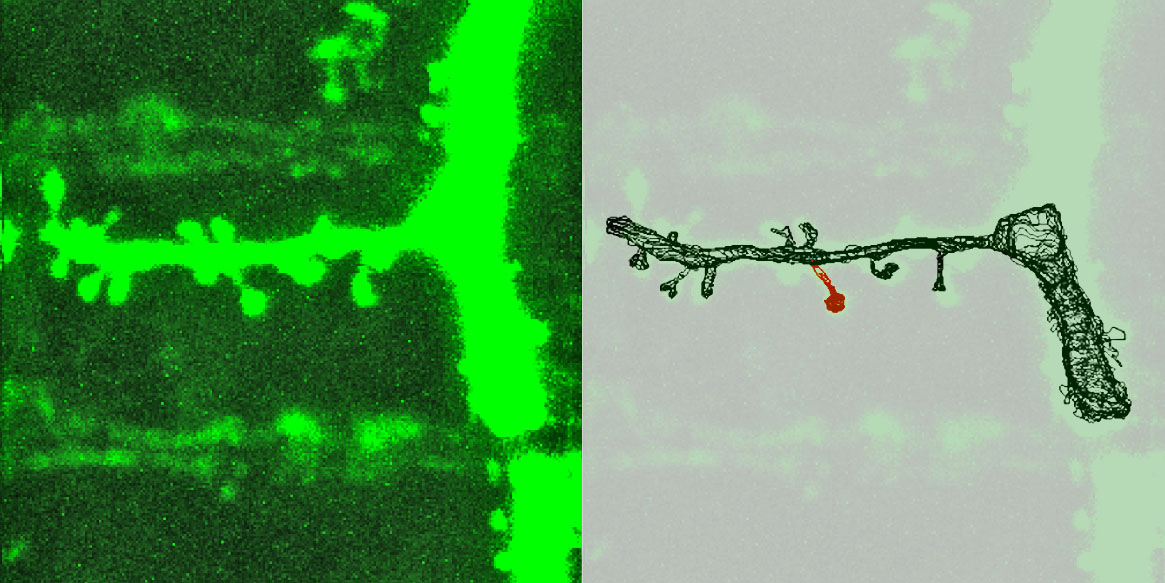
During the learning processes, extensions grow on neurons. Synapses are located at the end of these extensions (left: as seen in nature; right: reconstruction). (Credit: MPI of Neurobiology/ Meyer)
Researchers at the Max Planck Institute of Neurobiology in Martinsried have found an explanation for how a synapse stays stable for a long time despite the fact that its proteins must be renewed regularly.
The researchers studied the growth of synapses in tissue culture dishes following exposure to a learning stimulus. To do this, they activated individual synapses using the neurotransmitter glutamate, which plays an important role in learning processes and stimulates the growth of synapses. The researchers then observed the stimulated synapses and control synapses under a 2-photon microscope and electron microscope.
They discovered that during synapse growth, the different protein structures always grew coordinated with each other. If one structural component was enlarged alone, or in a way that was not correctly correlated with the other components, its structural change would collapse soon after. Synapses with such incomplete changes allow for forgetting things.
The study findings show that the order and interaction between synaptic components is finely tuned and correlated. “In a system of this kind, it should be entirely possible to replace individual proteins while the rest of the structure maintains its integrity,” says study leader Volker Scheuss.
The study’s results also establish a basis for a better understanding of memory loss, as in degenerative brain diseases.
Abstract of Neuron paper
- Spine, PSD, and bouton increase in concert after local glutamate application
- The amount of Homer1c increases within 20 min of spine enlargement
- The amount of PSD-95 increases after 80 min only in spines with stable enlargement
- The correlation in spine, PSD, and bouton size is a signature of synapse stability
Subsynaptic structures such as bouton, active zone, postsynaptic density (PSD) and dendritic spine, are highly correlated in their dimensions and also correlate with synapse strength. Why this is so and how such correlations are maintained during synaptic plasticity remains poorly understood. We induced spine enlargement by two-photon glutamate uncaging and examined the relationship between spine, PSD, and bouton size by two-photon time-lapse imaging and electron microscopy. In enlarged spines the PSD-associated protein Homer1c increased rapidly, whereas the PSD protein PSD-95 increased with a delay and only in cases of persistent spine enlargement. In the case of nonpersistent spine enlargement, the PSD proteins remained unchanged or returned to their original level. The ultrastructure at persistently enlarged spines displayed matching dimensions of spine, PSD, and bouton, indicating their correlated enlargement. This supports a model in which balancing of synaptic structures is a hallmark for the stabilization of structural modifications during synaptic plasticity.