How to reprogram cancer cells back to normal
August 25, 2015
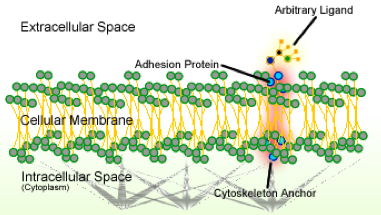
Schematic of cell adhesion (credit: Wikipedia)
A way to potentially reprogram cancer cells back to normalcy has been discovered by researchers on Mayo Clinic’s Florida campus.
The finding, published in Nature Cell Biology, represents “an unexpected new biology that provides the code, the software for turning off cancer,” says the study’s senior investigator, Panos Anastasiadis, Ph.D., chair of the Department of Cancer Biology on Mayo Clinic’s Florida campus.
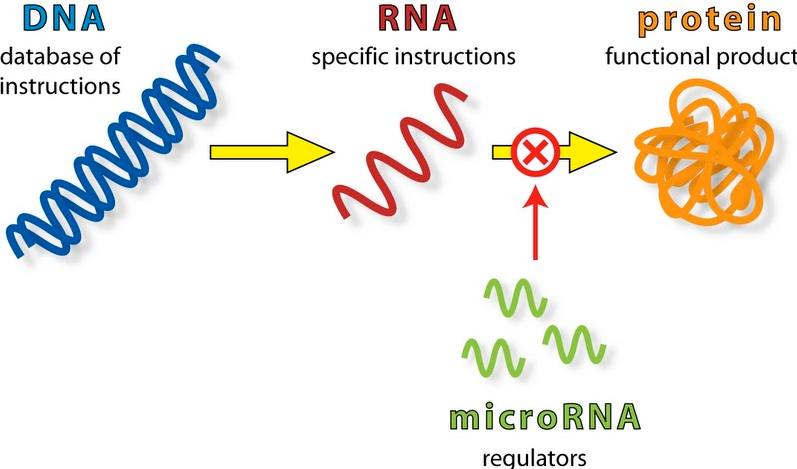
MicroRNAs — short, non-coding RNAs present in all living organisms that were formerly considered “junk DNA” — have been shown to regulate the expression of at least half of all human genes. These single-stranded RNAs exert their regulatory action by binding messenger RNAs and preventing their translation into proteins. (credit: Firefly BioWorks)
That code was unraveled by the discovery that adhesion proteins — the glue that keeps cells together — interact with a key player in the production of molecules called microRNAs (miRNAs).
The miRNAs orchestrate whole cellular programs by simultaneously regulating expression of a group of genes. The investigators found that when normal cells come in contact with each other, a specific subset of miRNAs suppresses (or blocks) genes that promote cell growth.
However, when adhesion is disrupted in cancer cells, these miRNAs are misregulated and cells grow out of control. The investigators showed, in laboratory experiments, that restoring normal miRNA levels in cancer cells can reverse that aberrant cell growth.
“The study brings together two so-far unrelated research fields — cell-to-cell adhesion and miRNA biology — to resolve a long-standing problem* about the role of adhesion proteins in cell behavior that was baffling scientists,” says the study’s lead author Antonis Kourtidis, Ph.D., a research associate in Anastasiadis’ lab. “Most significantly, it uncovers a new strategy for cancer therapy,” he adds.
“By administering the affected miRNAs in cancer cells to restore their normal levels, we should be able to re-establish the brakes and restore normal cell function,” Anastasiadis says. “Initial experiments in some aggressive types of cancer are indeed very promising.”
Mayo Clinic | Mayo Clinic Researchers Find New Code That Makes Reprogramming of Cancer Cells Possible
* That problem arose from conflicting reports about E-cadherin and p120 catenin — adhesion proteins that are essential for normal epithelial tissues to form, and which have long been considered to be tumor suppressors.
“However, we and other researchers had found that this hypothesis didn’t seem to be true, since both E-cadherin and p120 are still present in tumor cells and required for their progression,” Anastasiadis says. “That led us to believe that these molecules have two faces — a good one, maintaining the normal behavior of the cells, and a bad one that drives tumorigenesis.”
Their theory turned out to be true, but what was regulating this behavior was still unknown. To answer this, the researchers studied a new protein called PLEKHA7, which associates with E-cadherin and p120 only at the top, or the “apical” part of normal polarized epithelial cells. The investigators discovered that PLEKHA7 maintains the normal state of the cells, via a set of miRNAs, by tethering the microprocessor to E-cadherin and p120. In this state, E-cadherin and p120 exert their good tumor suppressor sides.
However, “when this apical adhesion complex was disrupted after loss of PLEKHA7, this set of miRNAs was misregulated, and the E-cadherin and p120 switched sides to become oncogenic,” Dr. Anastasiadis says.
“We believe that loss of the apical PLEKHA7-microprocessor complex is an early and somewhat universal event in cancer,” he adds. “In the vast majority of human tumor samples we examined, this apical structure is absent, although E-cadherin and p120 are still present. This produces the equivalent of a speeding car that has a lot of gas (the bad p120) and no brakes (the PLEKHA7-microprocessor complex).
Abstract of Distinct E-cadherin-based complexes regulate cell behaviour through miRNA processing or Src and p120 catenin activity
E-cadherin and p120 catenin (p120) are essential for epithelial homeostasis, but can also exert pro-tumorigenic activities. Here, we resolve this apparent paradox by identifying two spatially and functionally distinct junctional complexes in non-transformed polarized epithelial cells: one growth suppressing at the apical zonula adherens (ZA), defined by the p120 partner PLEKHA7 and a non-nuclear subset of the core microprocessor components DROSHA and DGCR8, and one growth promoting at basolateral areas of cell–cell contact containing tyrosine-phosphorylated p120 and active Src. Recruitment of DROSHA and DGCR8 to the ZA is PLEKHA7 dependent. The PLEKHA7–microprocessor complex co-precipitates with primary microRNAs (pri-miRNAs) and possesses pri-miRNA processing activity. PLEKHA7 regulates the levels of select miRNAs, in particular processing of miR-30b, to suppress expression of cell transforming markers promoted by the basolateral complex, including SNAI1, MYC and CCND1. Our work identifies a mechanism through which adhesion complexes regulate cellular behaviour and reveals their surprising association with the microprocessor.