Intricate microdevices that can be safely implanted
January 13, 2017

Fabrication and assembly of an iMEMS microdevice. Left: layer-by-layer fabrication of support structures and assembly of gear components. Right: the complete device after the layers have been sealed, with ferromagnetic iron material (black) to enable external magnetic control. (credit: SauYin Chin/Columbia Engineering)
Columbia Engineering researchers have invented a technique for manufacturing complex microdevices with three-dimensional, freely moving parts made from biomaterials that can safely be implanted in the body. Potential applications include a drug-delivery system to provide tailored drug doses for precision medicine, catheters, stents, cardiac pacemakers, and soft microbotics.
Most current implantable microdevices have static components rather than moving parts and, because they require batteries or other toxic electronics, they have limited biocompatibility.
The new technique stacks a soft biocompatible hydrogel material in layers, using a fast manufacturing method the researchers call “implantable microelectromechanical systems” (iMEMS).
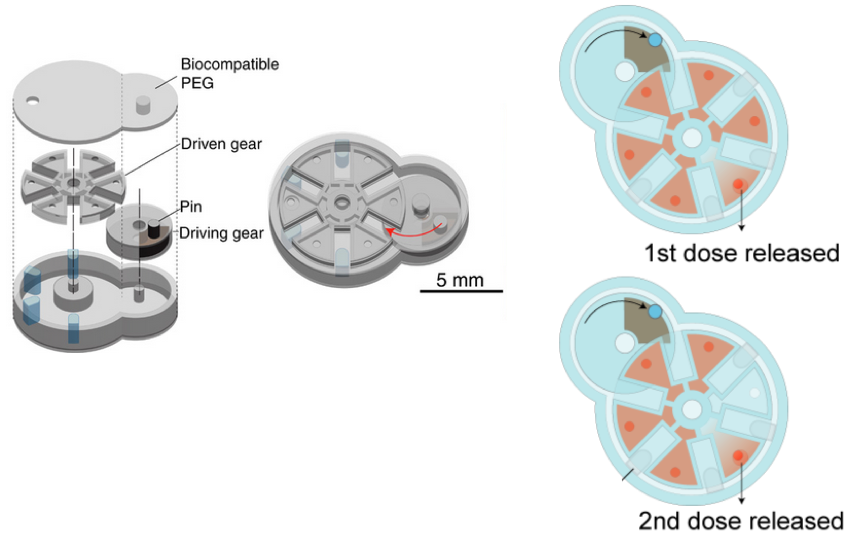
iMEMS drug-delivery system. The payload delivery system was tested in a bone cancer mouse model, finding that the triggering of releases of doxorubicin from the device over 10 days showed high treatment efficacy and low toxicity, at 1/10th of the standard systemic chemotherapy dose. The device contains iron nanoparticle–doped components, which respond to external magnetic actuation. Actuation of the device triggers release of payloads from reservoirs. (credit: Sau Yin Chin et al./Science Robotics)
“Our iMEMS platform enables development of biocompatible implantable microdevices with a wide range of intricate moving components that can be wirelessly controlled on demand, and solves issues of device powering and biocompatibility,” says Biomedical Engineering Professor Sam Sia, senior author of an open-access paper published online January 4, 2017, in Science Robotics).
The researchers were able to trigger the iMEMS device to release payloads over days to weeks after implantation, with precise actuation by using magnetic forces to induce gear movements that, in turn, bend structural beams made of hydrogels with highly tunable properties. (Magnetic iron particles are commonly used and are FDA-approved for human use as contrast agents.)
Batteryless implantable medical devices or sensors
Sia’s iMEMS technique addresses several issues in building biocompatible microdevices, micromachines, and microrobots: how to power small robotic devices without using toxic batteries; how to make small, biocompatible, moveable components that are not silicon, which has limited biocompatibility; and how to communicate wirelessly once implanted (radio-frequency microelectronics require power, are relatively large, and are not biocompatible).
The researchers developed a “locking mechanism” for precise actuation and movement of freely moving parts, which can function as valves, manifolds, rotors, pumps, and drug delivery systems. They were able to tune the biomaterials within a wide range of mechanical and diffusive properties and to control them after implantation without a sustained power supply, such as a toxic battery.
“We can make small implantable devices, sensors, or robots that we can talk to wirelessly. Our iMEMS system could bring the field a step closer to developing soft miniaturized robots that can safely interact with humans and other living systems,” said Sia.
The team developed a drug delivery system and tested it on mice with bone cancer. The iMEMS system delivered chemotherapy adjacent to the cancer, and limited tumor growth while showing less toxicity than with chemotherapy administered throughout the body.
The study was supported by the National Science Foundation, NIH, and the Agency for Science, Technology and Research (Singapore).
* The team used light to polymerize sheets of gel and incorporated a stepper mechanization to control the z-axis and pattern the sheets layer by layer, giving them three-dimensionality. Controlling the z-axis enabled the researchers to create composite structures within one layer of the hydrogel while managing the thickness of each layer throughout the fabrication process. They were able to stack multiple layers that are precisely aligned and, because they could polymerize a layer at a time, one right after the other, the complex structure was built in under 30 minutes.
“Hydrogels are difficult to work with, as they are soft and not compatible with traditional machining techniques,” says Sau Yin Chin, lead author of the study, who worked with Sia. “We have tuned the mechanical properties and carefully matched the stiffness of structures that come in contact with each other within the device. Gears that interlock have to be stiff in order to allow for force transmission and to withstand repeated actuation. Conversely, structures that form locking mechanisms have to be soft and flexible to allow for the gears to slip by them during actuation, while at the same time they have to be stiff enough to hold the gears in place when the device is not actuated. We also studied the diffusive properties of the hydrogels to ensure that the loaded drugs do not easily diffuse through the hydrogel layers.”
Abstract of Additive manufacturing of hydrogel-based materials for next-generation implantable medical devices
Implantable microdevices often have static components rather than moving parts and exhibit limited biocompatibility. This paper demonstrates a fast manufacturing method that can produce features in biocompatible materials down to tens of micrometers in scale, with intricate and composite patterns in each layer. By exploiting the unique mechanical properties of hydrogels, we developed a “locking mechanism” for precise actuation and movement of freely moving parts, which can provide functions such as valves, manifolds, rotors, pumps, and delivery of payloads. Hydrogel components could be tuned within a wide range of mechanical and diffusive properties and can be controlled after implantation without a sustained power supply. In a mouse model of osteosarcoma, triggering of release of doxorubicin from the device over 10 days showed high treatment efficacy and low toxicity, at 1/10 of the standard systemic chemotherapy dose. Overall, this platform, called implantable microelectromechanical systems (iMEMS), enables development of biocompatible implantable microdevices with a wide range of intricate moving components that can be wirelessly controlled on demand, in a manner that solves issues of device powering and biocompatibility.