MIT’s fast synthesis system could boost peptide-drug development
March 28, 2014
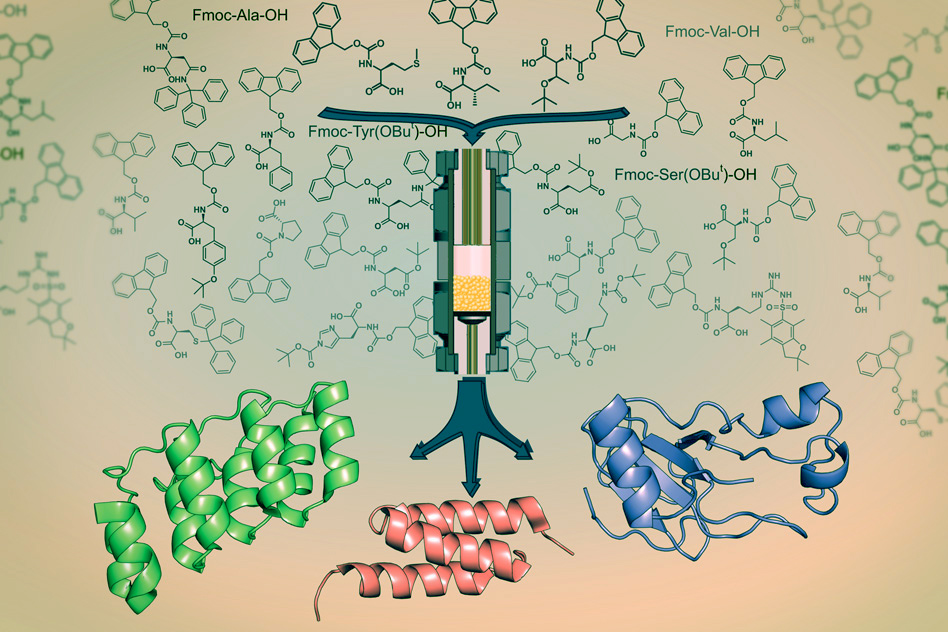
MIT chemists have devised a way to rapidly combine amino acids into protein fragments known as peptides for use in drugs (credit: Alexander Vinogradov)
Small protein fragments, also called peptides, are promising as drugs because they can be designed for very specific functions inside living cells, but manufacturing the peptides takes several weeks, making it difficult to obtain large quantities, and to rapidly test their effectiveness.
A team of MIT chemists and chemical engineers has designed a way to manufacture peptides in mere hours. The new system, described in a recent issue of the journal ChemBioChem, could have a major impact on peptide drug development, says Bradley Pentelute, an assistant professor of chemistry and leader of the research team.
“Peptides are ubiquitous. They’re used in therapeutics, they’re found in hydrogels, and they’re used to control drug delivery. They’re also used as biological probes to image cancer and to study processes inside cells,” Pentelute says. “Because you can get these really fast now, you can start to do things you couldn’t do before.”
Insulin and the HIV drug Fuzeon are some of the earliest successful examples, and peptide drugs are expected to become a $25 billion market by 2018, the researchers say.
Accelerated manufacturing
Therapeutic peptides usually consist of a chain of 30 to 40 amino acids, the building blocks of proteins. Many universities, including MIT, have facilities to manufacture these peptides, but the process usually takes two to six weeks, using machines developed about 20 years ago. These machines require about an hour to perform the chemical reactions needed to add one amino acid to a chain.
To speed up the process, the MIT team adapted the synthesis reactions so they could be done in a continuous flow system. Using this approach, each amino acid addition takes only a few minutes, and an entire peptide can be assembled in little more than an hour.
In future versions, “we think we’re going to be able to do each step in under 30 seconds,” says Pentelute, who is also an associate member of the Broad Institute. “What that means is you’re really going to be able to do anything you want in short periods of time.”
The new system has storage vessels for each of the 20 naturally occurring amino acids, connected to pumps that pull out the correct one. As the amino acids flow toward the chamber where the reaction takes place, they travel through a coil where they are preheated to 60 degrees Celsius, which helps speed up the synthesis reaction.
This system produces peptides as pure as those produced with existing machines. “We’re on par with the world’s best state-of-the-art synthesis, but we can do it much faster now,” Pentelute says.
The system should make it easier to design and produce new peptides, says Philip Dawson, an associate professor of chemistry at the Scripps Research Institute who was not part of the research team. “What I like about it is the simplicity of the device and the thoughtfulness with which it was put together,” Dawson says. “I would look forward to trying it out in my lab.”
With this technology, scientists could design and rapidly test new peptides to treat cancer and other diseases, as well as more effective variants of existing peptides, such as insulin, Pentelute says. Another benefit of this high-speed approach is that any potential problems with a particular peptide synthesis can be detected much sooner, allowing the researchers to try to fix it right away.
Another area Pentelute plans to pursue is creating so-called “mirror-image” proteins. Nearly all proteins that exist in nature are made of L amino acids, whose structures have a right-handed orientation. Creating and studying peptides that are mirror images of these natural proteins could pave the way to developing such peptides as new drugs with completely different functions from the right-handed versions.
Making large synthetic proteins
In a separate paper published in the same issue of ChemBioChem, the researchers demonstrated that they could use this technology not only to synthesize peptides, but also combine these to form large synthetic proteins. To demonstrate the technology, they created an antibody mimic that has 130 amino acids, as well as a 113-amino-acid enzyme produced by bacteria.
The researchers have patented the technology, and MIT’s Deshpande Center for Technological Innovation has given them a grant to help commercialize it. Pentelute says he believes that about 10 machines using the new technology would be enough to meet current demand, which is about 100,000 to 500,000 custom peptides per year.
Pentelute envisions that the technology could have an impact on synthetic biology comparable to rapid synthesis of short strands of DNA and RNA. These strands, known as oligonucleotides, take only a day or two to prepare and can be used to create custom genes to give cells new functions.
“That’s what our aim is — to have next-day or two-day delivery of these peptide units, to anyone in the world. That’s really the dream,” he says.
The research was funded by the MIT Reed Fund, the Deshpande Center, a Damon-Runyon-Rachleff Innovation Award, a Sontag Foundation Distinguished Scientist Award, a C.P. Chu and Y. Lai Fellowship, an AstraZeneca Distinguished Graduate Student Fellowship, the National Institute of General Medical Sciences, and the National Institutes of Health.
Abstract of ChemBioChem paper
A flow-based solid-phase peptide synthesis methodology that enables the incorporation of an amino acid residue every 1.8 min under automatic control or every 3 min under manual control is described. This is accomplished by passing a stream of reagent through a heat exchanger into a low volume, low backpressure reaction vessel, and through a UV detector. These features enable continuous delivery of heated solvents and reagents to the solid support at high flow rate, thereby maintaining maximal concentration of reagents in the reaction vessel, quickly exchanging reagents, and eliminating the need to rapidly heat reagents after they have been added to the vessel. The UV detector enables continuous monitoring of the process. To demonstrate the broad applicability and reliability of this method, it was employed in the total synthesis of a small protein, as well as dozens of peptides. The quality of the material obtained with this method is comparable to that for traditional batch methods, and, in all cases, the desired material was readily purifiable by RP-HPLC. The application of this method to the synthesis of the 113-residue Bacillus amyloliquefaciens RNase and the 130-residue DARPin pE59 is described in the accompanying manuscript.