Nano-enhanced textiles clean themselves with light
March 25, 2016

Close-up of nanostructures grown on cotton textiles. Image magnified 150,000 times. (credit: RMIT University)
Researchers at at RMIT University in Australia have developed a cheap, efficient way to grow special copper- and silver-based nanostructures on textiles that can degrade organic matter when exposed to light.
Don’t throw out your washing machine yet, but the work paves the way toward nano-enhanced textiles that can spontaneously clean themselves of stains and grime simply by being put under a light or worn out in the sun.
The nanostructures absorb visible light (via localized surface plasmon resonance — collective electron-charge oscillations in metallic nanoparticles that are excited by light), generating high-energy (“hot”) electrons that cause the nanostructures to act as catalysts for chemical reactions that degrade organic matter.
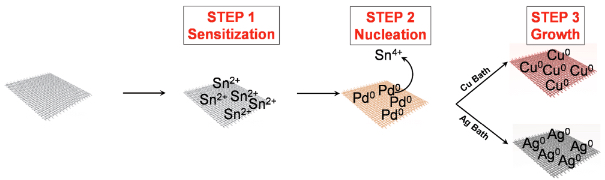
Steps involved in fabricating copper- and silver-based cotton fabrics: 1. Sensitize the fabric with tin. 2. Form palladium seeds that act as nucleation (clustering) sites. 3. Grow metallic copper and silver nanoparticles on the surface of the cotton fabric. (credit: Samuel R. Anderson et al./Advanced Materials Interfaces)
The challenge for researchers has been to bring the concept out of the lab by working out how to build these nanostructures on an industrial scale and permanently attach them to textiles. The RMIT team’s novel approach was to grow the nanostructures directly onto the textiles by dipping them into specific solutions, resulting in development of stable nanostructures within 30 minutes.
When exposed to light, it took less than six minutes for some of the nano-enhanced textiles to spontaneously clean themselves.
The research was described in the journal Advanced Materials Interfaces.
Scaling up to industrial levels
Rajesh Ramanathan, a RMIT postdoctoral fellow and co-senior author, said the process also had a variety of applications for catalysis-based industries such as agrochemicals, pharmaceuticals, and natural products, and could be easily scaled up to industrial levels. “The advantage of textiles is they already have a 3D structure, so they are great at absorbing light, which in turn speeds up the process of degrading organic matter,” he said.

Cotton textile fabric with copper-based nanostructures. The image is magnified 200 times. (credit: RMIT University)
“Our next step will be to test our nano-enhanced textiles with organic compounds that could be more relevant to consumers, to see how quickly they can handle common stains like tomato sauce or wine,” Ramanathan said.
“There’s more work to do to before we can start throwing out our washing machines, but this advance lays a strong foundation for the future development of fully self-cleaning textiles.”
Abstract of Robust Nanostructured Silver and Copper Fabrics with Localized Surface Plasmon Resonance Property for Effective Visible Light Induced Reductive Catalysis
Inspired by high porosity, absorbency, wettability, and hierarchical ordering on the micrometer and nanometer scale of cotton fabrics, a facile strategy is developed to coat visible light active metal nanostructures of copper and silver on cotton fabric substrates. The fabrication of nanostructured Ag and Cu onto interwoven threads of a cotton fabric by electroless deposition creates metal nanostructures that show a localized surface plasmon resonance (LSPR) effect. The micro/nanoscale hierarchical ordering of the cotton fabrics allows access to catalytically active sites to participate in heterogeneous catalysis with high efficiency. The ability of metals to absorb visible light through LSPR further enhances the catalytic reaction rates under photoexcitation conditions. Understanding the modes of electron transfer during visible light illumination in Ag@Cotton and Cu@Cotton through electrochemical measurements provides mechanistic evidence on the influence of light in promoting electron transfer during heterogeneous catalysis for the first time. The outcomes presented in this work will be helpful in designing new multifunctional fabrics with the ability to absorb visible light and thereby enhance light-activated catalytic processes.