Neural probes for the spinal cord
April 6, 2017
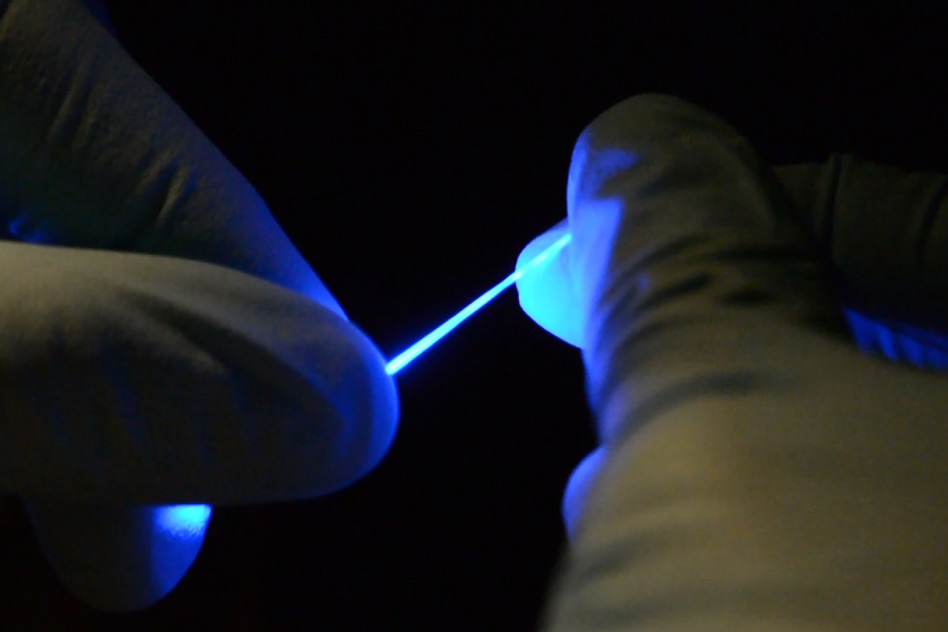
Researchers have developed a rubber-like fiber, shown here, that can flex and stretch while simultaneously delivering both optical impulses for optoelectronic stimulation,and electrical connections for stimulation and monitoring. (credit: Chi (Alice) Lu and Seongjun Park)
A research team led by MIT scientists has developed rubbery fibers for neural probes that can flex and stretch and be implanted into the mouse spinal cord.
The goal is to study spinal cord neurons and ultimately develop treatments to alleviate spinal cord injuries in humans. That requires matching the stretchiness, softness, and flexibility of the spinal cord. In addition, the fibers have to deliver optical impulses (for optoelectronic stimulation of neurons with blue or yellow laser light) and have electrical connections (for electrical stimulation and monitoring of neurons).
Implantable fibers have allowed brain researchers to stimulate specific targets in the brain and monitor electrical responses. But similar studies in the nerves of the spinal cord have been more difficult to carry out. That’s because the spine flexes and stretches as the body moves, and the relatively stiff, brittle fibers used today could damage the delicate spinal cord tissue.
The scientists used a newly developed elastomer (a tough elastic polymer material that can flow and be stretched) that is transparent (like a fiber optic cable) for transmitting optical signals, and formed an external mesh coating of silver nanowires as a conductive layer for electrical signals. Think of it as tough, transparent, silver spaghetti.
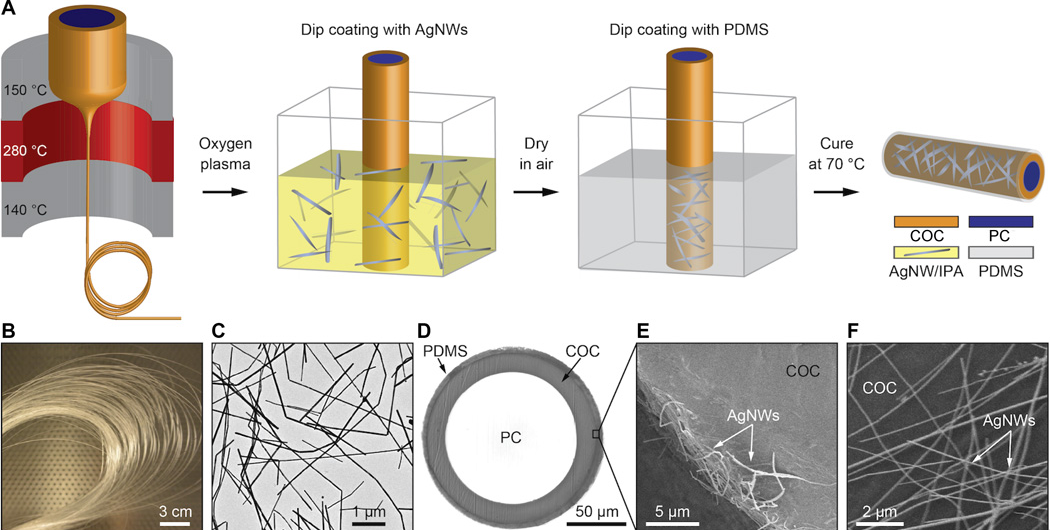
Fabrication of flexible neural probes. (A) Thermal (heat) drawing produced a flexible optical fiber that also served as a structural core for the probe. (B) Spool of a fiber transparent polycarbonate (PC) core and cyclic olefin copolymer (COC) cladding, which enabled the fiber to be drawn into a fiber and was dissolved away after the drawing process. (C) Transmission electron microscopy (TEM) image of silver nanowires (AgNW). (D) Cross-sectional image of the fiber probe with biocompatible polydimethylsiloxane (PDMS) coating. (E) Scanning electron microscopy image showing a portion of the ring silver nanowire electrode cross section. (F) Scanning electron microscopy image of the silver nanowire mesh on top of the fiber surface. (credit: Chi Lu et al./Science Advances)
The fibers are “so floppy, you could use them to do sutures and deliver light at the same time,” says MIT Professor Polina Anikeeva. The fiber can stretch by at least 20 to 30 percent without affecting its properties, she says. “Eventually, we’d like to be able to use something like this to combat spinal cord injury. But first, we have to have biocompatibility and to be able to withstand the stresses in the spinal cord without causing any damage.”
Scientists doing research on spinal cord injuries or disease usually must use larger animals in their studies, because the larger nerve fibers can withstand the more rigid wires used for stimulus and recording. While mice are generally much easier to study and available in many genetically modified strains, there was previously no technology that allowed them to be used for this type of research.
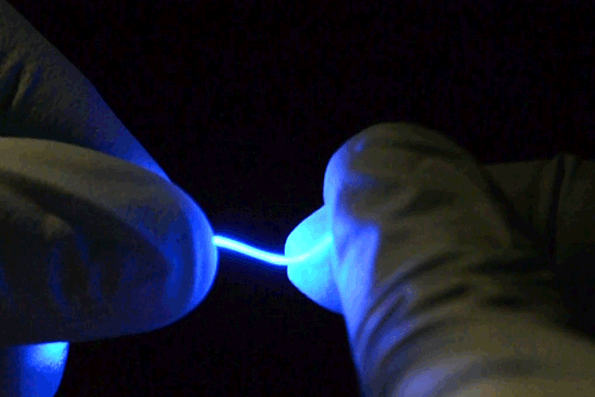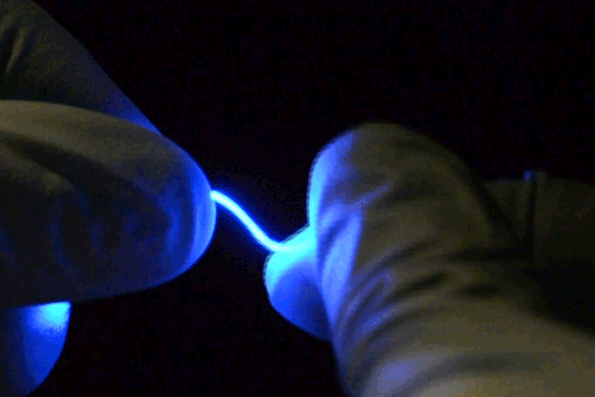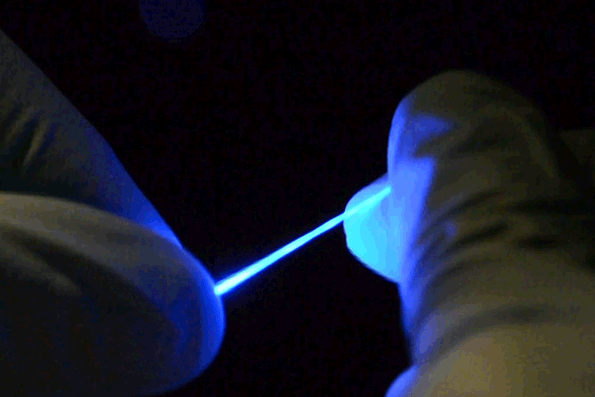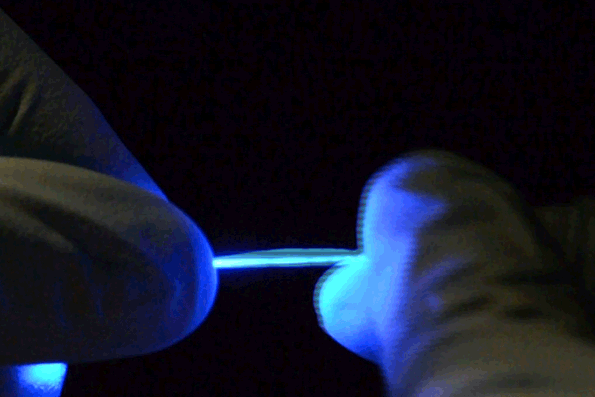
The fibers are not only stretchable but also very flexible. (credit: Chi (Alice) Lu and Seongjun Park)
The team included researchers at the University of Washington and Oxford University. The research was supported by the National Science Foundation, the National Institute of Neurological Disorders and Stroke, the U.S. Army Research Laboratory, and the U.S. Army Research Office through the Institute for Soldier Nanotechnologies at MIT.
Abstract of Flexible and stretchable nanowire-coated fibers for optoelectronic probing of spinal cord circuits
Studies of neural pathways that contribute to loss and recovery of function following paralyzing spinal cord injury require devices for modulating and recording electrophysiological activity in specific neurons. These devices must be sufficiently flexible to match the low elastic modulus of neural tissue and to withstand repeated strains experienced by the spinal cord during normal movement. We report flexible, stretchable probes consisting of thermally drawn polymer fibers coated with micrometer-thick conductive meshes of silver nanowires. These hybrid probes maintain low optical transmission losses in the visible range and impedance suitable for extracellular recording under strains exceeding those occurring in mammalian spinal cords. Evaluation in freely moving mice confirms the ability of these probes to record endogenous electrophysiological activity in the spinal cord. Simultaneous stimulation and recording is demonstrated in transgenic mice expressing channelrhodopsin 2, where optical excitation evokes electromyographic activity and hindlimb movement correlated to local field potentials measured in the spinal cord.