New nanomaterial mimics cell membranes
July 20, 2016
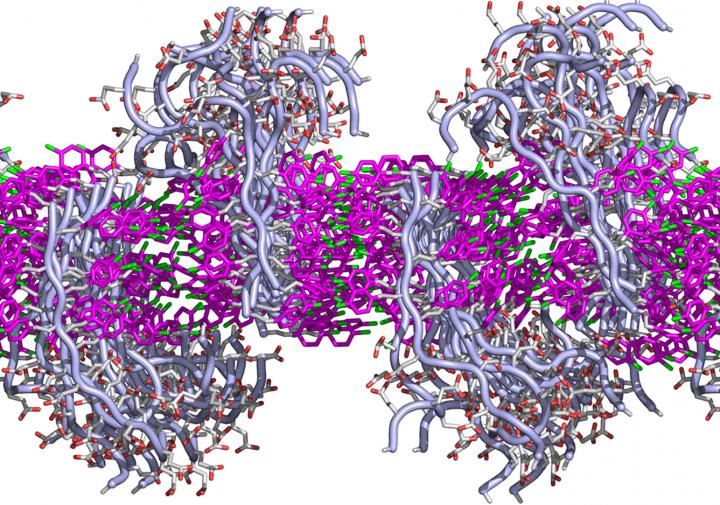
This simulated cross-section shows how the lipid-like peptoids interact to form a membrane. Each peptoid has two sections: a fatty-like region that interacts via benzene rings (shown in pink) with its neighbors to form a sheet, and a water-loving region that juts above or below the flat sheet. Each region can be designed to have specific functions. (credit: Chun-Long Chen/PNNL)
Materials scientists at the Department of Energy’s Pacific Northwest National Laboratory have created a new material that performs like a biological cell membrane — a material that has long been sought for applications like water purification and drug delivery.
The “peptoid” material can assemble itself into a sheet that’s thinner, but more stable, than a soap bubble, the researchers report this week in Nature Communications. The assembled sheet can withstand being submerged in a variety of liquids and can even repair itself after damage.
“We believe these materials have potential in water filters, sensors, drug delivery, and especially fuel cells or other energy applications,” said chemist Chun-Long Chen.
Biological cell membranes, which are made from thin sheets of fatty molecules called lipids, are at least ten times thinner than an iridescent soap bubble and yet allow cells to collectively form organisms as diverse at bacteria, trees and people.
Cell membranes are also very selective about what they let pass through, using tiny embedded proteins as gatekeepers. Membranes repair dings to their structure automatically and change thickness to pass signals from the outside environment to the cell’s interior, where most of the action is.
Scientists would like to take advantage of these gatekeeping and other membrane properties to make filters. A cell-membrane-like material would have advantages over other thin materials such as graphene. For example, mimicking a cell membrane’s efficient gatekeeping could result in water purifying membranes that don’t require a lot of pressure or energy to push the water through.
How to design imitation biological cell membranes
Synthetic molecules called peptoids have caught the interest of researchers because they are cheap, versatile and customizable. They are like natural proteins, including those that embed themselves in cell membranes, and can be designed to have very specific forms and functions. So Chen and colleagues decided to see if they could design peptoids to make them more lipid-like (that is, more like fats).
Lipid molecules are long and mostly straight: They have a fatty end that prefers to hang out with other fats, and a water-like end that prefers the comfort of water. Because of this chemistry, lipid molecules arrange themselves with the fatty ends pointed toward each other, sandwiched between the water-loving ends pointed out. Scientists call this a lipid bilayer, essentially a sheet that envelops the contents of a cell. Proteins or carbohydrate molecules embed themselves in the membranous sheet.
Inspired by this, Chen and colleagues designed peptoids in which each base peptoid was a long molecule with one end water-loving and the other end fat-loving. They chose chemical features that they hoped would encourage the individual molecules to pack together. They examined the resulting structures using a variety of analysis methods, including some at the Advanced Light Source and the Molecular Foundry, two DOE Office of Science User Facilities at Lawrence Berkeley National Laboratory.
Forming nanomembranes
The team found that after putting the lipid-like peptoids into a liquid solution, the molecules spontaneously crystallized and formed “nanomembranes” — straight-edged sheets as thin as cell membranes — floating in the beaker. These nanomembranes maintained their structure in water or alcohol, at different temperatures, in solutions with high or low pH, or high concentrations of salts, a feat that few cell membranes could accomplish.
To better understand the nanomembranes, the team simulated how single peptoid molecules interacted with each other using molecular dynamics software. The simulated peptoids formed a membrane reminiscent of a lipid bilayer: The fat-loving ends lined up in the middle, and their water-loving ends pointed outward either above or below.
They also confirmed the ability of the synthetic membranes to hold proteins that have specific functions, such as ones that let water, and only water, through, and to repair themselves.
The results showed the researchers that they are on the right path to making synthetic cell membrane-like materials. The next step, Chen said, is to build biomimetic membranes by incorporating natural membrane proteins or other synthetic water channels such as carbon nanotubes into these sheet matrices. The team is also looking into ways to make the peptoid membranes conductive for energy uses.
This work was supported by the Department of Energy Office of Science and PNNL.
Abstract of Highly stable and self-repairing membrane-mimetic 2D nanomaterials assembled from lipid-like peptoids
An ability to develop sequence-defined synthetic polymers that both mimic lipid amphiphilicity for self-assembly of highly stable membrane-mimetic 2D nanomaterials and exhibit protein-like functionality would revolutionize the development of biomimetic membranes. Here we report the assembly of lipid-like peptoids into highly stable, crystalline, free-standing and self-repairing membrane-mimetic 2D nanomaterials through a facile crystallization process. Both experimental and molecular dynamics simulation results show that peptoids assemble into membranes through an anisotropic formation process. We further demonstrated the use of peptoid membranes as a robust platform to incorporate and pattern functional objects through large side-chain diversity and/or co-crystallization approaches. Similar to lipid membranes, peptoid membranes exhibit changes in thickness upon exposure to external stimuli; they can coat surfaces in single layers and self-repair. We anticipate that this new class of membrane-mimetic 2D nanomaterials will provide a robust matrix for development of biomimetic membranes tailored to specific applications.