‘Normal’ bacteria vital for keeping intestinal lining intact, preventing disorders
August 20, 2014
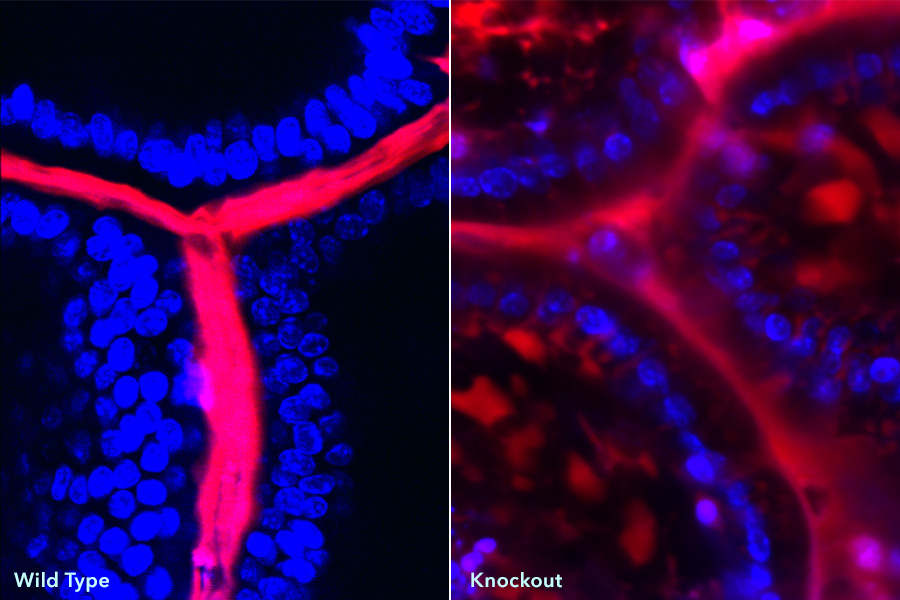
These images show intestines of wild-type and knockout mice injected with dextran (red) and imaged using two-photon microscopy. DAPI (blue) shows stained cells within the intestinal epithelium. Dye tracking (red) between DAPI (blue) labelled cells indicates a leaky intestinal epithelium. (Credit: Kamal Khanna, Ph.D., University of Connecticut, Farmington)
Bacteria that aid in digestion keep the intestinal lining intact, scientists at Albert Einstein College of Medicine of Yeshiva University and associates have found.
The findings, reported online in the journal Immunity, could yield new therapies for inflammatory bowel disease (IBD) and a wide range of other disorders.
The research involved the intestinal microbiome, which contains some 100 trillion bacteria. The role of these microorganisms in promoting or preventing disease is a major emerging field of study.
Einstein scientists found that absorption of a specific healthy bacterial byproduct is crucial for maintaining the integrity of the intestinal epithelium — the single-cell layer responsible for keeping intestinal bacteria and their toxins inside the gut and away from the rest of the body. Breaching of the intact intestinal epithelium is associated with a number of diseases.
“Intestinal bacteria secrete a wide variety of chemicals known as metabolites,” said Sridhar Mani, M.D., co-corresponding author of the paper. “These bacteria and their metabolites were known to influence the intestinal epithelium’s integrity, but precisely how they did so wasn’t known.” Mani is professor of medicine and of genetics and the Miriam Mandel Faculty Scholar in Cancer Research at Einstein and attending physician, oncology at the Montefiore Einstein Center for Cancer Care and Montefiore Medical Center.
How probiotics (healthy bacteria) restore the barrier
Mani and his colleagues suspected that bacterial metabolites exert their influence by binding to and activating a protein in the nuclei of intestinal epithelial cells called the pregnane X receptor (PXR). PXR was known to be activated by chemicals within the body (such as bile acids) as well as by drugs including steroids and antibiotics.
In a series of mouse studies, the researchers found that a metabolite called indole 3-propionic acid (IPA) — produced exclusively by “commensal” bacteria (where one organism benefits without affecting the other), which aid in digestion — both strengthens the intestinal epithelium’s barrier function and prevents its inflammation by activating PXR. More specifically, PXR activation suppresses production of an inflammatory protein called tumor necrosis factor alpha (TNF-α) while increasing levels of a protein that strengthens the junctions between adjacent intestinal epithelial cells.
Novel probiotics
“By adding probiotics in the form of IPA-producing bacteria to the intestine or by administering IPA directly, we may be able to prevent or treat IBD and other inflammatory disorders that occur when the intestinal epithelium has been compromised,” said Dr. Mani. “Such a strategy could also be tried for other health problems that may occur when the intestinal epithelium breaks down, including certain forms of liver disease, diabetes, asthma, allergies, obesity and heart disease.”
Dr. Mani’s team is now developing novel probiotics aimed at restoring the intestinal epithelium’s barrier function by encouraging IPA’s interaction with PXR.
The research was supported by National Institutes of Health grants and a Damon Runyon Foundation Clinical Investigator Award.
Abstract of Immunity paper
- Pregnane X receptor (PXR) is a physiologic regulator of intestinal permeability
- Microbial-derived indoles can regulate intestinal barrier function through PXR
- PXR regulates intestinal barrier function through TLR4
- Specific indole-producing bacteria can reduce intestinal inflammation
Intestinal microbial metabolites are conjectured to affect mucosal integrity through an incompletely characterized mechanism. Here we showed that microbial-specific indoles regulated intestinal barrier function through the xenobiotic sensor, pregnane X receptor (PXR). Indole 3-propionic acid (IPA), in the context of indole, is a ligand for PXR in vivo, and IPA downregulated enterocyte TNF-α while it upregulated junctional protein-coding mRNAs. PXR-deficient (Nr1i2−/−) mice showed a distinctly “leaky” gut physiology coupled with upregulation of the Toll-like receptor (TLR) signaling pathway. These defects in the epithelial barrier were corrected in Nr1i2−/−Tlr4−/− mice. Our results demonstrate that a direct chemical communication between the intestinal symbionts and PXR regulates mucosal integrity through a pathway that involves luminal sensing and signaling by TLR4.