Possible biochemical mechanism underlying long-term memories identified
December 4, 2015
It’s a nagging question: why do some of our memories fade away, while others last forever? Now scientists at the Stowers Institute for Medical Research have identified a possible biochemical mechanism: a specific synaptic protein called Orb2 can either block or maintain neural synapses (connections between neurons), which create and maintain long-term memories.
So for a memory to persist, the synaptic connections must be kept strong. But how? The researchers previously identified a synaptic protein called CPEB that is responsible for maintaining the strength of such connections in the sea slug (a model organism used in memory research). Recently, they identified a similar protein, called Orb2, in the fruit fly.
Now, using a fruit fly model system, they found that the synaptic connections are kept strong by the transformation of Orb2 from one molecular state to another. And that transformation causes Orb2 molecules to solidify and strengthen the memory connections in the brain.
The authors conclude their paper, published in the current issue of the journal Cell, with several questions. How and what triggers this transformation, how long does it persist? Is the continued presence of a prion-like state necessary for the persistence of memory, and is it correlated with or predictive of long-lasting memory? And most interestingly: can a transient memory about to be forgotten be stabilized by artificial recruitment of the prion-like state (perhaps by a neurotropic compound)?
And what about that ironic link with prions, associated with neurodegenerative disorders? Are prions some twisted form of memory that could one day even have value? We’ll be keeping an eye on where this fascinating research leads.
Technical details: the memory switch
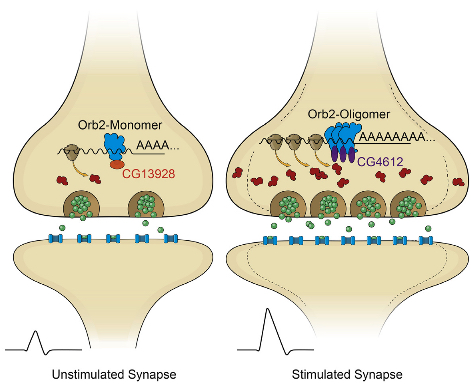
In their latest study, the researchers determined that Orb2 exists in two distinct physical states: monomeric (a single molecule that can bind to other molecules) and oligomeric (a molecular complex).
Like CPEB, oligomeric Orb2 is prion-like — that is, it’s a self-copying cluster. (But unlike prions, oligomeric Orb2 and CPEB are not toxic.) Monomeric Orb2 represses, and oligomeric Orb2 activates a crucial step in the complex cellular process that leads to protein synthesis.
During this crucial step, messenger RNA (mRNA), which is an RNA copy of a gene’s recipe for a protein, is translated by the cell’s ribosome into the sequence of amino acids that will make up a newly synthesized protein. The monomeric form of Orb2 binds to the target mRNA, keeping it in a repressed state.
The Stowers scientists also determined that prion-like Orb2 not only activates translation into amino acids but imparts its translational state to nearby monomer forms of Orb2. As a result, monomeric Orb2 is transformed into prion-like Orb2, so its role in translation switches from repression to activation.
Self-sustaining activation maintains synaptic activity
Stowers Associate Investigator Kausik Si, Ph.D. thinks this switch is the possible mechanism by which fleeting experiences create an enduring memory. “Because of the self-sustaining nature of the prion-like state, this creates a local and self-sustaining translation activation of Orb2-target mRNA, which maintains the changed state of synaptic activity over time,” says Si.
The discovery that the two distinct states of Orb2 have opposing roles in the translation process provides “for the first time a biochemical mechanism of synapse-specific persistent translation and long-lasting memory,” he states.
“To our knowledge, this is the first example of a prion-based protein switch that turns a repressor into an activator,” Si adds. “The recruitment of distinct protein complexes at the non-prion and prion-like forms to create altered activity states indicates the prion-like behavior is in essence a protein conformation-based switch.
“Through this switch, a protein can lose or gain a function that can be maintained over time in the absence of the original stimuli. Although such a possibility has been anticipated prior to this study, there was no direct evidence.”
The research builds upon previous studies by Si and Eric Kandel, M.D., of Columbia University and other scientists. These studies revealed that both short-term and long-term memories are created in synapses.
Abstract of Amyloidogenic Oligomerization Transforms Drosophila Orb2 from a Translation Repressor to an Activator
Memories are thought to be formed in response to transient experiences, in part through changes in local protein synthesis at synapses. In Drosophila, the amyloidogenic (prion-like) state of the RNA binding protein Orb2 has been implicated in long-term memory, but how conformational conversion of Orb2 promotes memory formation is unclear. Combining in vitro and in vivo studies, we find that the monomeric form of Orb2 represses translation and removes mRNA poly(A) tails, while the oligomeric form enhances translation and elongates the poly(A) tails and imparts its translational state to the monomer. The CG13928 protein, which binds only to monomeric Orb2, promotes deadenylation, whereas the putative poly(A) binding protein CG4612 promotes oligomeric Orb2-dependent translation. Our data support a model in which monomeric Orb2 keeps target mRNA in a translationally dormant state and experience-dependent conversion to the amyloidogenic state activates translation, resulting in persistent alteration of synaptic activity and stabilization of memory.