Reprogramming neurons and rewiring the brain
November 16, 2015
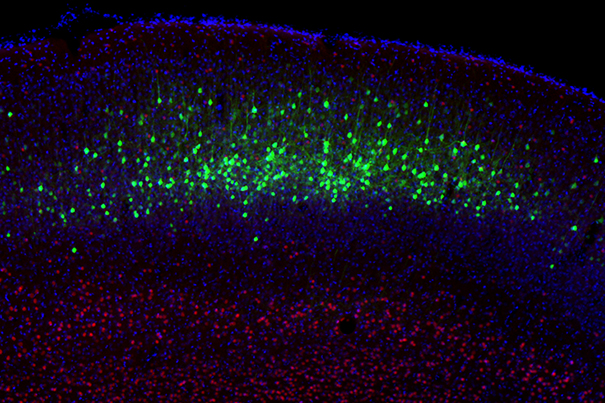
Neurons of the cerebral cortex targeted for reprogramming are displayed in green. (credit: Caroline Rouaux/Arlotta Lab)
In previous research, Harvard Stem Cell Institute researchers disproved neurobiology dogma by “reprogramming” neurons — turning one form of neuron into another — in the brains of living animals. Now they’ve taken it a step further, showing that networks of communication among reprogrammed neurons and their neighbors can also be changed, or “rewired.”
The finding, by Paola Arlotta, a professor of stem cell and regenerative biology, in close collaboration with Takao Hensch, a professor of molecular and cellular biology, has implications for both a basic understanding of how neurons choose their synaptic partners when circuits wire during early development, and for developing strategies to change defective communication in psychiatric and neurodevelopmental diseases, such as schizophrenia and autism, the researchers suggest.
In the new research, the research team reprogrammed one type of excitatory neurons into another type of excitatory neurons, and then observed connections made by a type of inhibitory neurons, noticing that the inhibitory neurons then recognized the reprogrammed excitatory neurons as “new” cells and modified their circuitry as a result.*
“What we’ve shown here is that not only neurons can be dramatically changed from one type into another from within the brain,” said Arlotta, but also that “neighboring neurons recognize the reprogrammed cells as different and adapt by changing how they communicate with them. Transformed neurons were recognized as ‘new’ cells, with different properties, by neighboring inhibitory interneurons, which in turn created new circuitry appropriate for their ‘new’ neuronal target,” Arlotta said.
That demonstrates that “synaptic connections among neurons are not made randomly,” she added. “The brain is much more sophisticated, and different neurons have ways to control the behavior of neighboring circuits in their own unique way to ultimately change how much inhibition, for example, they receive from their synaptic partners.”
Rewiring the brain to fix a damaged circuit
One of the major focuses today in regenerative neurobiology is to use stem cells to produce and replace neurons killed off by disease, and then implant them into patients’ brains. “Instead of making neurons in a dish from stem cells, an alternative would be to reprogram the identity of other neurons,” Arlotta said.
“These are early but exciting days,” Arlotta said. “The work thus far has been done in the brains of young mice, which are far more plastic than adult brains,” she pointed out. The next frontier, she said, is to attempt to reprogram neurons and circuits in older brains. “If we could do it in an adult brain, it would be immensely powerful.”
Arlotta is also an associate member of the Stanley Center for Psychiatric Disease at the Broad Institute of Harvard and MIT.
The work is published in the latest edition of the journal Neuron.
* The researchers first reprogrammed “glutamatergic excitatory projection neurons (PNs)” (neurons of the cerebral cortex that normally would connect the two sides of the brain) into another type of PN neurons that instead connect far away, for example in the spinal cord. They then monitored the behavior of a third class of cells, “local GABAergic inhibitory interneurons (INs)” and showed that the INs change their synaptic connections to connect to the reprogrammed PN neurons.
Abstract of Instructing Perisomatic Inhibition by Direct Lineage Reprogramming of Neocortical Projection Neurons
During development of the cerebral cortex, local GABAergic interneurons recognize and pair with excitatory projection neurons to ensure the fine excitatory-inhibitory balance essential for proper circuit function. Whether the class-specific identity of projection neurons has a role in the establishment of afferent inhibitory synapses is debated. Here, we report that direct in vivo lineage reprogramming of layer 2/3 (L2/3) callosal projection neurons (CPNs) into induced corticofugal projection neurons (iCFuPNs) increases inhibitory input onto the converted neurons to levels similar to that of endogenous CFuPNs normally found in layer 5 (L5). iCFuPNs recruit increased numbers of inhibitory perisomatic synapses from parvalbumin (PV)-positive interneurons, with single-cell precision and despite their ectopic location in L2/3. The data show that individual reprogrammed excitatory projection neurons extrinsically modulate afferent input by local PV+ interneurons, suggesting that projection neuron class-specific identity can actively control the wiring of the cortical microcircuit.