Temperature-sensitive gel scaffold regenerates craniofacial bone
December 13, 2013
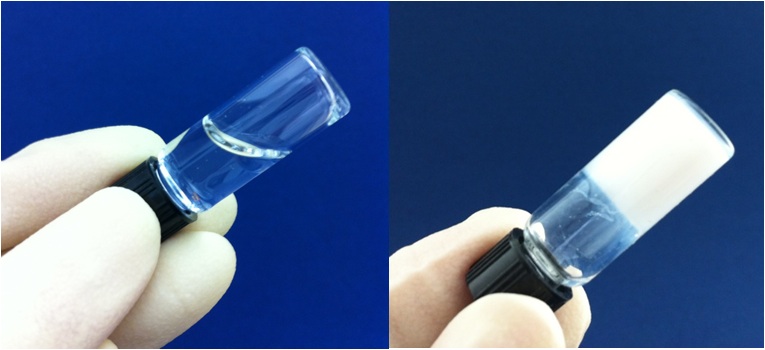
Injectable hydrogel scaffold starts as a soluble liquid at room temperature, left, and forms a stable, nonshrinking gel, right, at body temperature after one minute (credit: Mikos Laboratory/Rice University)
Rice University bioengineers have developed a hydrogel scaffold for regeneratomg craniofacial bone tissue. The hydrogel starts as a liquid, solidifies into a gel in the body, and liquefies again for removal.
The material developed in the Rice lab of bioengineer Antonios Mikos is a soluble liquid at room temperature that can be injected where needed. At body temperature, the material turns instantly into a gel to help direct the formation of new bone to replace that damaged by injury or disease. It also serves as an effective delivery vehicle for growth factors and stem cell populations.
The gel conforms to irregular 3D spaces and provides a platform for functional and aesthetic tissue regeneration. It is intended as an alternative to prefabricated implantable scaffolds.
“This new platform technology leverages injectable, thermally responsive, chemically crosslinkable and bioresorbable hydrogels for regenerative medicine applications,” Mikos said. “It enables the formation of scaffolds locally and the delivery of growth factors and stem cells into defects of complex anatomical shapes with minimal surgical intervention.”
Once sufficient quality and quantity of bone tissue have regenerated to fill the defected site, the hydrogel scaffold can be transitioned back into a liquid state and released naturally.
As part of the project, the researchers will test the hydrogel’s enhanced seeding capabilities and ability to promote cellular attachment, crosstalk and proliferation toward greater bone formation. The knowledge will improve the understanding of biomaterial-based therapies for minimally invasive tissue regeneration as viable clinical alternatives.
“The results demonstrate the ability to encapsulate stem cell populations with temperature-sensitive gelling scaffolds for injectable cell delivery with enormous implications for the development of novel therapeutics for craniofacial bone regeneration,” Mikos said.
The National Institutes of Health, the Baylor College of Medicine Scientific Training Program for Dental Academic Researchers and the Kirschstein fellowship supported the research.
Abstract of Biomacromolecules paper
Injectable, dual-gelling hydrogels were successfully developed through the combination of physical thermogellation at 37°C and favorable amine:epoxy chemical crosslinking. Poly(N-isopropylacrylamide)-based thermogelling macromers with a hydrolyzable lactone ring and epoxy pendant groups, and a biodegradable diamine-functionalized polyamidoamine crosslinker were synthesized, characterized, and combined to produce non-syneresing and bioresorbable hydrogels. Differential scanning calorimetry and oscillatory rheometry demonstrated the rapid and dual-gelling nature of the hydrogel formation. The post-gelation dimensional stability, swelling, and mechanical behavior of the hydrogel system were shown to be easily tuned at the synthesis and formulation stages. The leachable products were found to be cytocompatible at all conditions, while the degradation products demonstrated a dose- and time-dependent response due to solution osmolality. Preliminary encapsulation studies showed MSC viability could be maintained for 7 days. The results suggest that injectable, thermally and chemically crosslinkable hydrogels are promising alternatives to prefabricated biomaterials for tissue engineering applications, particularly for cell delivery.