The BRAIN mapping initiative needs rethinking
April 10, 2013 by Don Stein
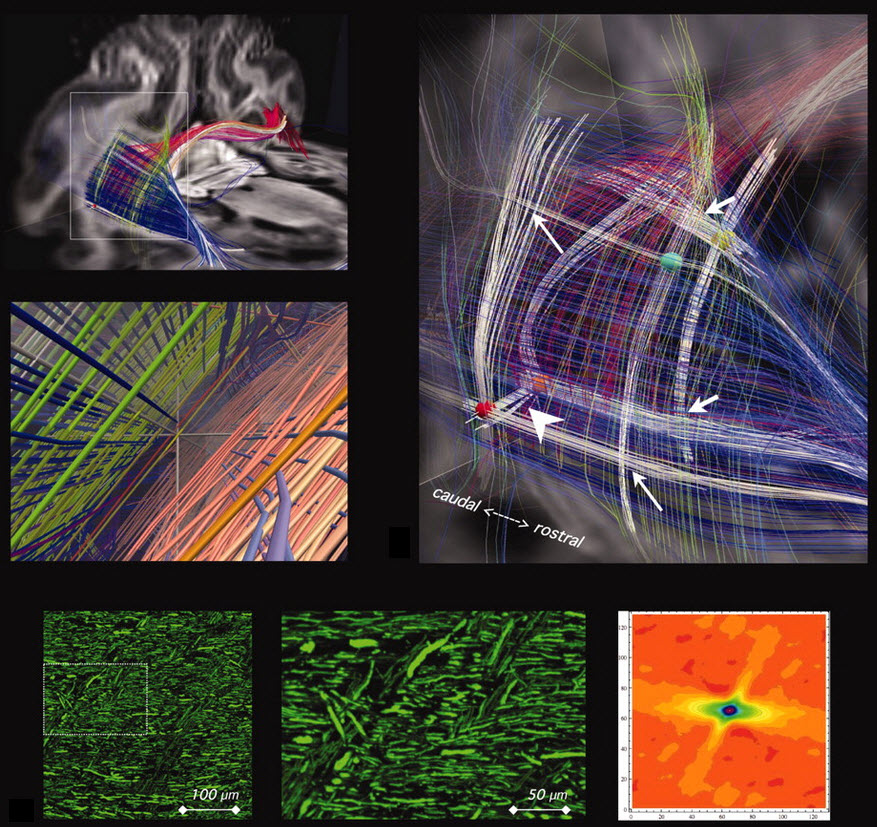
Grid structure of cerebral pathways (credit: Science)
As a biomedical research scientist I am concerned about President Obama’s broad new research initiative “to map the human brain.”
The BRAIN ((Brain Research through Advancing Innovative Neurotechnologies) initiative is a very ambitious, and perhaps even noble, effort, and I am most definitely not against imaging or nanotechnology as tools for research.
But, without specific goals, hypotheses or endpoints, the research effort becomes a fishing expedition. That is, if we throw enough technology at the project and get enough people involved, something is sure to come of it — maybe.
I am also not against Big Science projects, if they are based on viable precepts. However, I do think we need to have a more thoughtful discussion of the immediate and long-term issues, with a wider range of participants and perspectives, and some attention to alternatives and priorities, before we dedicate increasingly limited, long-term public funding to such an effort — starting with $100 million per year and a proposed rise to at least $300 million per year for at least 15 years.
Senior scientists in the president’s administration have compared the brain-mapping initiative to the human genome project, but in a recent New York Times article, John Markoff and James Gorman rightly pointed out that, “It is different however, in that it has, as yet, no clearly defined goals or endpoint.”
In a subsequent interview with Jonathan Hamilton on National Public Radio, the director of the National Institutes of Health, Francis Collins, made the same point. In an article last month, also in the New York Times, Tim Requarth pointed out: “Other critics say the project is too open-ended — that it makes little sense without clearly defined criteria for success. ‘It’s not like the Human Genome Project, where you just have to read out a few billion base pairs and you’re done,’ said Peter Dayan, a neuroscientist at University College London. ‘For the human brain, what would you need to know to build a simulation? That’s a huge research question, and it has to do with what’s important to know about the brain.'”
Every scientist (including me) would love to be able to get a grant without having to specify any goals, hypotheses or endpoints, but is this a realistic way to do science?
Why is this mapping initiative more important than other possible initiatives? Is it more important than finding a cure for AIDS? More relevant than beating cancer in all its manifestations? Although the notion of mapping everything going on in the brain has curb appeal, such an open-ended endeavor calls for at least some solid evidence that it is likely to produce substantive changes in disease outcome, understanding of diseases and better public health for the nation.
Consciousness Raising for neuroscience
A deep problem hampering this discussion is the near-universal lack of awareness about the limited, historically determined, very probably transient character of our prevailing assumptions about the relationship between gray matter and brain function.
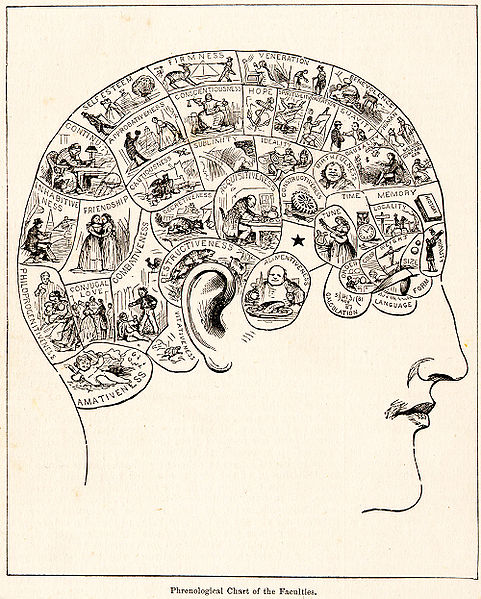
An 1883 phrenology chart (credit: People’s Cyclopedia of Universal Knowledge/Wikimedia Commons)
The attraction of brain mapping owes much to an obsolete scientific paradigm. Attempts to map and parcellate the human (and animal) brain into morphologically and anatomically distinct areas, each with its specific function, have been around for more than a century.
In the mid-1800s it became scientifically fashionable in neurology to discover and “map” the functions of the cerebral cortex using a variety of methods and techniques available at any given moment. This was called phrenology, and this mapping paradigm became the major focus of the neurological disciplines that led to the doctrine of cerebral localization of functions.
The phrenological trend continues to the present; its ever more sophisticated technologies mask what some of us consider an obsolescent concept (the article by Cold Spring Harbor Laboratory professor Partha Mitra in Scientific American presented a good example).
Mapping the brain with modern technology is a direct extension of that same paradigm. The paramount question here is not about the technology per se, but whether what it represents and what it measures is an accurate reflection what we want to know about how the brain works. Given what we’ve learned so far, we have to ask whether the concept is valid or whether we are calling for a lot of effort and spending based on an outmoded paradigm.
Is mapping a valid concept?
Although it is well established that the connections between dendrites and synapses in the brain are in a state of constant change, we cannot seem to get out from under the idea that brain activity has some kind of shape — a geography that lines up with function. The brain does not sleep and nothing ever gets turned off in the brains of living creatures.
The map of what connects to what must always be changing. Any one instant of imaging will represent just that instant and perhaps nothing more. A map of how “billions, if not trillions of nerve cells interact” has also to account for the role of the billions upon billions of support cells called glia that also make up the brain. No one associated with the mapping initiative seems to be asking what these critical cells contribute to normal and abnormal functions of the brain — so the dynamics and dynamical changes that are always in flux are not going to be characterized by temporally static or even dynamic measures, no matter how technologically sophisticated they may be.
“Maps” are, at absolute best, only limited approximations of the constantly intense dynamics of brain activity, structure and function. The neuroscience community cannot agree as to what it is, exactly, that should be mapped. Molecular changes? Genomic changes? Proteins? Structural changes? Electrical? Biochemical? All of those “events” involve vast numbers of signaling pathways, each of which affects the others in a vibrant, ever-changing cascade. And this doesn’t even begin to address how environmental and behavioral feedback loops affect these mechanisms.
In the present state of neuroscience, there is no consensus about the best approach to mapping, and which approaches should be given the highest priority. And as Mithra notes, even if we could map the action potentials for every single neuron in the mammalian brain, how do we make the jump to complex behavior that emerges from the measurement of action potentials? When, and for how long, will recording have to be done to generate that information?
This is no small issue. Others have also expressed concern that current imaging technologies have often been incorrectly applied, leading to the wrong conclusions about how the brain is “wired” and how it functions in a dynamic state.
How should we proceed?
Before we try to map brains (even brains of worms and fruit flies and mice), we need to work out better concepts of what needs to be measured, and then apply the appropriate technologies to measure it. As it now stands, we have high-level technology with no clear concept of what to measure and no defined goals or endpoints. Does the project simply go on forever? When will we know that we have the answers? I agree with others that despite the rhetoric of administration spokespersons and those who will benefit directly, this is not at all like the genome or moon-landing projects.
In my own field of specialization, traumatic brain injury and stroke, we know that even humans with massive damage to the brain can make remarkable recoveries of function — under the right conditions — sometimes almost instantly. The problem we face is how to unlock those conditions. Brain maps cannot account for this extensive plasticity and repair at all, any more than most diseases can be attributed to the regulation and expression of just one gene — as most systems biologists will tell you if given the chance.
What practical outcomes do we expect?
Some have argued that investing in the mapping project will generate new jobs and wealth, and this could happen. The Human Genome Project is generating considerable wealth and biomedical startup companies (for example, screening genomes for individual clients) — certainly more than the initial dollars invested. However, the actual benefit to patients has so far been very limited. We now know a lot about the human genome map, but how many diseases have been cured?
New York Times reporter Gina Kolata, recently reporting on DNA testing for rare disorders, noted that sequencing of the entire genome of patients with rare diseases is becoming so popular that the costs are now down, from $7,000 to $9,000 for a family, and demand is soaring — hence the commercial value of such tests. Yet all the sequencing offers no panacea, she says: “Genetic aberrations are only found in about 25 percent of cases, less then 3 percent get better management of their disease and only about 1 percent get an actual treatment and a major benefit.”
With the Brain Mapping Initiative, are we about to make a very heavy investment in a project that promises no end-points and nothing specific in the way of actual benefit? If so, we ought to be clear about it and not let the public think that “miraculous cures” and full understanding of brain functions are just around the corner.
We need to talk
I urge that we need broader and more considered discussion of how we want to invest our research resources. I marvel that a small group of scientists were able to catch the president’s attention and support, but is this kind of earmarking in lieu of salient peer-review the way we want to decide allocations for research? We hate it when Congress does this (if we’re not the beneficiaries), so do we want to adopt the same model? These questions should all be a part of the debate.
Whether I agree with the paradigm or not, I most certainly support those who still want to continue research on brain mapping. But we need to look again at whether it merits the disproportionate investment and prestige proposed for it, especially now, in a time of severe, perhaps permanent curtailments in biomedical research funding.
This is not about big science or small science and this is not just about the $100 million kick-start — the stakes and costs will be much higher. This is about good science and bad science, or at best, not-so-good science. In the current zero-sum game of funding, many other areas of critical biomedical research, including hundreds of small, or smaller, projects with potential for important near-term clinical application, will suffer as the money goes elsewhere and as students and researchers flock to where the money is. Is this good for biomedical research? Are we sure?