Tracking large neural networks in the brain by making neurons glow
November 30, 2016
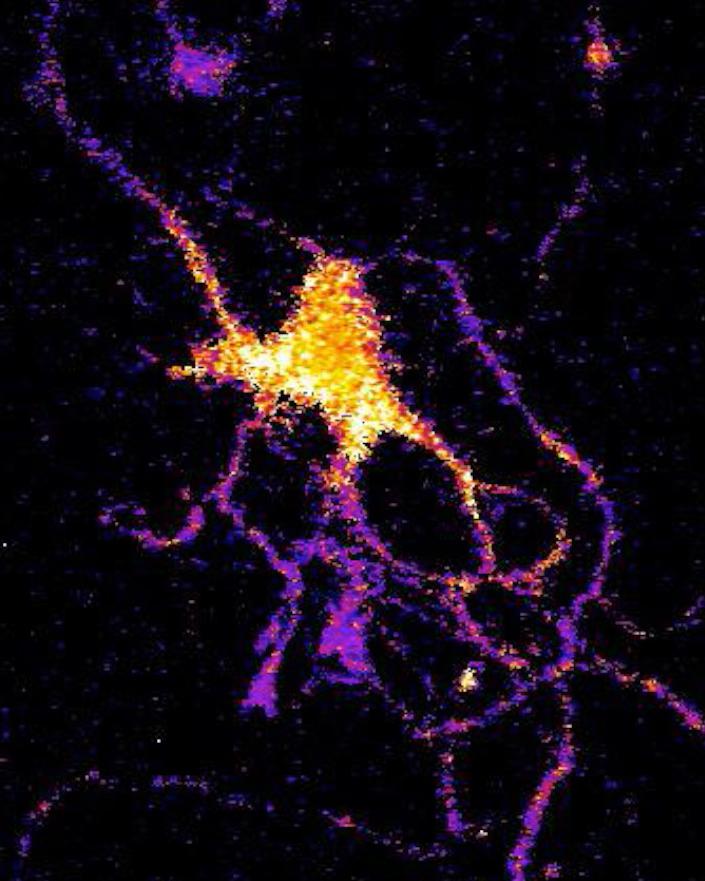
A neuron glows with bioluminescent light produced by a new genetically engineered sensor. (credit: Johnson Lab, Vanderbilt University)
A new kind of bioluminescent sensor developed by Vanderbilt scientists causes individual brain cells to glow in the dark, giving neuroscientists a new tool to track what’s happening in large neural networks in the brain.
The sensor is a genetically modified form of luciferase, the enzyme that fireflies and other species use to produce light.
Traditional electrical techniques for recording the activity of neurons are limited to small numbers of neurons at a time. Instead, the new sensors use a new combination of optical techniques to record the activity of hundreds of neurons at the same time, according to Carl Johnson, Stevenson Professor of Biological Sciences, who headed the effort.
The research is a spinoff of the team’s research in bioluminescence* in the green alga Chlamydomonas. Johnson and his colleagues realized that if they could combine luminescence with optogenetics (which uses light to control cells, particularly neurons), they could create a powerful new tool for studying brain activity.
To create the new sensor, Johnson and his collaborators first genetically modified a type of luciferase obtained from a luminescent species of shrimp so that it would light up when exposed to calcium ions (their level pikes briefly when a neuron receives an impulse from one of its neighbors). Then they attached a virus that infects and genetically modifies neurons.
They tested their new calcium sensor with an optogenetic protein called channelrhodopsin that causes the calcium ion channels in the neuron’s outer membrane to open, flooding the cell with calcium. Using neurons grown in culture they found that the luminescent enzyme reacted visibly to the influx of calcium produced when the probe was stimulated by brief light flashes of visible light.
Vanderbilt University | Bioluminescent sensor causes brain cells to glow in the dark
To determine how well their sensor works with larger numbers of neurons, they inserted it into brain slices from mouse hippocampus containing thousands of neurons. In this case, they flooded the slices with an increased concentration of potassium ions, which causes the cell’s ion channels to open. Again, they found that the sensor responded to the variations in calcium concentrations by brightening and dimming.
Vanderbilt University | Optical sensor illuminates activity of neural network
“We’ve shown that the approach works,” Johnson said. “Now we have to determine how sensitive it is. We have some indications that it is sensitive enough to detect the firing of individual neurons, but we have to run more tests to determine if it actually has this capability.”
The research is described in a paper published in the open-access journal Nature Communications on Oct. 27 and was funded by the National Institutes of Health, the National Science Foundation, and a grant from the Vanderbilt Brain Institute.
* There have also been efforts in optical recording of neurons using fluorescence, but this requires a strong external light source which can cause the tissue to heat up and can interfere with some biological processes, particularly those that are light sensitive, Johnson explained. It would also interfere with the light used in optogenetics. In contrast, light in bioluminescence is produced by biochemical reactions (the light-emitting pigment luciferin and the enzyme luciferase).
Abstract of Coupling optogenetic stimulation with NanoLuc-based luminescence (BRET) Ca++ sensing
Optogenetic techniques allow intracellular manipulation of Ca++ by illumination of light-absorbing probe molecules such as channelrhodopsins and melanopsins. The consequences of optogenetic stimulation would optimally be recorded by non-invasive optical methods. However, most current optical methods for monitoring Ca++ levels are based on fluorescence excitation that can cause unwanted stimulation of the optogenetic probe and other undesirable effects such as tissue autofluorescence. Luminescence is an alternate optical technology that avoids the problems associated with fluorescence. Using a new bright luciferase, we here develop a genetically encoded Ca++ sensor that is ratiometric by virtue of bioluminescence resonance energy transfer (BRET). This sensor has a large dynamic range and partners optimally with optogenetic probes. Ca++ fluxes that are elicited by brief pulses of light to cultured cells expressing melanopsin and to neurons-expressing channelrhodopsin are quantified and imaged with the BRET Ca++ sensor in darkness, thereby avoiding undesirable consequences of fluorescence irradiation.