Triggered by ultrasound, microbubbles open the blood-brain barrier to administer drugs without harming other areas of the body
December 2, 2016
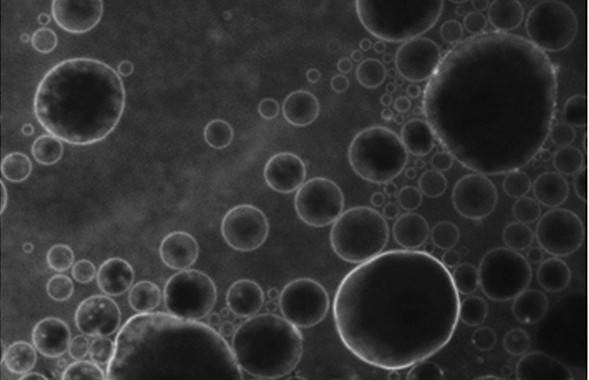
Microbubbles containing a new fluorescent substance in their lipid coating, released in a designated point in the brain by ultrasound (credit: C. Sierra et al./Columbia University UEIL)
Using ultrasound to bypass the blood-brain barrier (BBB), Columbia University researchers have succeeded in releasing drugs only in the specific area of the brain where they are needed — not in the rest of the body. The goal is to help treat Parkinson’s, Alzheimer’s, and other neurodegenerative diseases without collateral damage.
The BBB is an impassable obstacle for 98% of drugs, which it treats as pathogens and blocks them from passing from patients’ bloodstream into the brain. Using ultrasound (sound whose frequency is higher than the range of human hearing), drugs can administered using an intravenous injection of innocuous lipid-coated gas microbubbles. That technique was perfected by Columbia University scientists, who reported their research in 2011 in PNAS and in 2014 in the Journal of Cerebral Blood Flow & Metabolism.
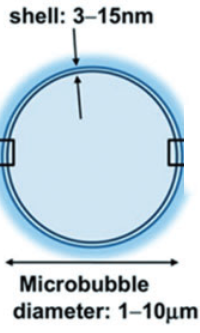
Microbubble (credit: C. Sierra et al./Journal of Cerebral Blood Flow & Metabolism)
With this method, ultrasound is focused on a specific region of the brain, causing the microbubbles to oscillate and increase in size and expand. When they reach the critical size of 8 microns, the blood–brain barrier near them opens, allowing the medicine circulating in the blood to pass through.
This technique has been used experimentally for over ten years, but has had a disadvantage: at excessive pressure, the microbubbles can suddenly collapse, so there is no lipid shell to retain the covering of the microbubbles, allowing the contained drug to flow through the blood pool and causing microdamage to the cerebral vessels and elsewhere.
Now, scientists at the Ultrasound Elasticity Imaging Laboratory (UEIL) at Columbia University have taken a major step forward by incorporating a fluorescent molecule called 5-dodecanoylaminofluorescein into the lipid coating of the microbubbles.
This molecule allows the scientists to determine the optimal pressure to prevent bursting of the microbubbles*, according to UEIL physicist Carlos Sierra and lead author of a paper on this new finding published in the current issue of Journal of Cerebral Blood Flow & Metabolism.
Human trials planned
So far, the researchers have proven the efficacy of their technique on mice, confirming that this molecule was reaching the brain without affecting other parts of the animal. They also identified the acoustic pressure thresholds at which the substance is guaranteed to safely reach its target in vivo.
“Defining these parameters means we can think about how to transfer the technique to human patients, although it has to be tested on monkeys first,” Sierra explains. “It could be applied to diseases like Parkinson’s, Alzheimer’s, Huntington’s diseases, brain tumors, strokes, multiple sclerosis, and amyotrophic lateral sclerosis, where we expect to see a very significant rise in the efficacy of treatment and a considerable reduction in side effects.”
Sierra is funded by a grant from Berrié Foundation in Spain.
* By ex vivo fluorescence imaging and by in vivo transcranial passive cavitation detection.
Abstract of Lipid microbubbles as a vehicle for targeted drug delivery using focused ultrasound-induced blood–brain barrier opening
Focused ultrasound in conjunction with lipid microbubbles has fully demonstrated its ability to induce non-invasive, transient, and reversible blood–brain barrier opening. This study was aimed at testing the feasibility of our lipid-coated microbubbles as a vector for targeted drug delivery in the treatment of central nervous system diseases. These microbubbles were labeled with the fluorophore 5-dodecanoylaminfluorescein. Focused ultrasound targeted mouse brains in vivo in the presence of these microbubbles for trans-blood–brain barrier delivery of 5-dodecanoylaminfluorescein. This new approach, compared to previously studies of our group, where fluorescently labeled dextrans and microbubbles were co-administered, represents an appreciable improvement in safety outcome and targeted drug delivery. This novel technique allows the delivery of 5-dodecanoylaminfluorescein at the region of interest unlike the alternative of systemic exposure. 5-dodecanoylaminfluorescein delivery was assessed by ex vivo fluorescence imaging and by in vivo transcranial passive cavitation detection. Stable and inertial cavitation doses were quantified. The cavitation dose thresholds for estimating, a priori, successful targeted drug delivery were, for the first time, identified with inertial cavitation were concluded to be necessary for successful delivery. The findings presented herein indicate the feasibility and safety of the proposed microbubble-based targeted drug delivery and that, if successful, can be predicted by cavitation detection in vivo.