Turning skin cells directly into cells that insulate neurons
April 17, 2013
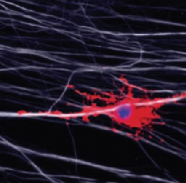
Skin cell transformed into an oligodendrocyte precursor cell (OPC) (red), which myelinates (covers and insulates) the axon of a nerve cell (credit: Nan Yang et al./Nature Biotechnology)
Researchers at the Stanford University School of Medicine have succeeded in transforming skin cells directly into oligodendrocyte precursor cells (OPCs), the cells that myelinate nerve cells (wrap them in the insulating myelin sheaths that help nerve signals propagate) and would work successfully when transplanted into the brains of mice with a myelin disorder.
The current research was done in mice and rats. If the approach also works with human cells, it could eventually lead to cell therapies for diseases like inherited leukodystrophies — disorders of the brain’s white matter — and multiple sclerosis, as well as spinal cord injuries.
Background: repairing myelin disorders
Without myelin to insulate neurons, signals sent down nerve cell axons quickly lose power. Diseases that attack myelin, such as multiple sclerosis, result in nerve signals that are not as efficient and cannot travel as far as they should.
Myelin disorders can affect nerve signal transmission in the brain and spinal cord, leading to cognitive, motor and sensory problems.
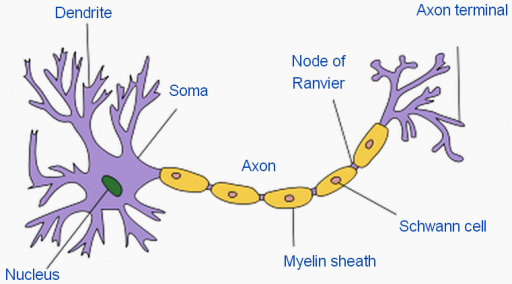
Structure of a typical neuron, showing myelin sheath (credit: Wikimedia Commons)
Previous research in rodent disease models has shown that transplanted oligodendrocyte precursor cells derived from embryonic stem cells and from human fetal brain tissue can successfully create myelin sheaths around nerve cells, sometimes leading to dramatic improvements in symptoms.
Creating OPCs from skin cells directly
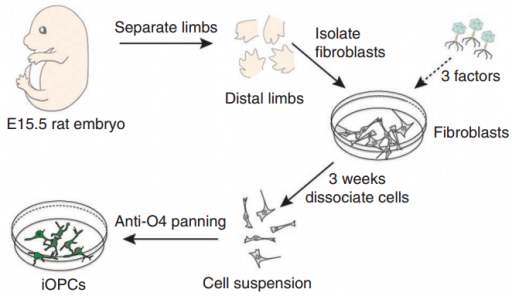
Converting embryonic skin cells (fibroblasts) directly into induced oligodendrocyte precursor cells (iOPCs) (credit: Nan Yang et al./Nature Biotechnology)
“Unfortunately, the availability of human fetal tissue is extremely limited, and the creation of OPCs from embryonic stem cells is slow and tedious,” said the study’s senior author, Marius Wernig, MD, assistant professor of pathology and a member of Stanford’s Institute for Stem Cell Biology and Regenerative Medicine. “It appeared we wouldn’t be able to create enough human OPCs for widespread therapeutic use, so we began to wonder if we could create them directly from skin cells.”
Last year, Wernig’s team successfully created human nerve cells out of embryonic skin cells. Other researchers had successfully used a similar process to turn skin cells into embryonic-like cells called induced pluripotent stem cells, and then grow those iPS cells into nerve cells, but Wernig’s lab was the first to convert embryonic skin cells (fibroblasts) directly into nerve cells without the intermediate iPS cell step.
The team’s current research project also involved directly converting skin cells without the intermediate iPS cell step, but into induced OPCs (iOPCs) instead of nerve cells.
Nan Yang, PhD, a postdoctoral scholar in the Wernig laboratory and lead author of the study, pointed out that there is another advantage to using this technique. “By using the patient’s own skin cells, we should be able to generate transplantable OPCs that are genetically identical to the patient’s natural OPCs,” Yang said. “This allows us to avoid the problem of immune rejection, which is a major complication in transplantation medicine.”
Human cells next
Next, the team plans to reproduce the research in human cells; if successful, the approach could lay the groundwork for therapies for a wide array of myelin disorders and spinal cord injuries.
The research was supported by the Ellison Medical Foundation, the Stinehart-Reed Foundation, the National Institutes of Health, the Howard Hughes Medical Institute, the Swedish Research Council, the Swedish Society for Medical Research, and the New York Stem Cell Foundation. Stanford’s Department of Pathology and Institute for Stem Cell Biology and Regenerative Medicine also supported the work.
Stanford University Medical Center integrates research, medical education and patient care at its three institutions: Stanford University School of Medicine, Stanford Hospital & Clinics and Lucile Packard Children’s Hospital.