Ultrathin ‘diagnostic skin’ allows continuous patient monitoring
January 2, 2014

The array laminates without adhesives onto the surface of the skin by soft contact much like a temporary transfer tattoo. Contact is maintained despite normal stretching or pinching of the skin. (Credit: John Rogers, University of Illinois at Urbana-Champaign)
An entirely new approach to measuring body temperature — an ”electronic skin” that adheres non-invasively to human skin, conforms well to contours, and provides a detailed temperature map of any surface of the body — has been developed by an international multidisciplinary team including researchers at the University of Illinois at Urbana/Champaign and the National Institute of Biomedical Imaging and Bioengineering (NIBIB).
Subtle variations in temperature can indicate potentially harmful underlying conditions such as constriction or dilation of blood vessels, or dehydration. Even changes in mental activity, such as increased concentration while solving a mathematical equation, are accompanied by measureable changes in body temperature.
Detecting skin temperature changes can serve as early indicators of disease development and progression. For example, sophisticated infrared digital cameras can detect, in high resolution, temperature changes across large areas of the body. At the other end of the technology spectrum, paste-on temperature sensors provide simple, single-point measurements. Although both technologies are accurate, infrared cameras are expensive and require the patient to remain completely still, and while paste-on sensors allow free movement, they provide limited information.
How it works
The temperature sensor array is a variation of a novel technology, originally developed in the lab of Professor John Rogers at the University of Illinois at Urbana/Champaign, called “epidermal electronics,” consisting of ultrathin, flexible skin-like arrays that contain sensors and heating elements. The arrays resemble a tattoo of a micro-circuit board.
The technology offers the potential for a wide range of diagnostic and therapeutic capabilities with little patient discomfort. For example, sensors can be incorporated that detect different metabolites of interest. Similarly, the heaters can be used to deliver heat therapy to specific body regions; actuators can be added that deliver an electrical stimulus or even a specific drug. Future versions will have a wireless power coil and an antenna for remote data transfer.
Testing the new device
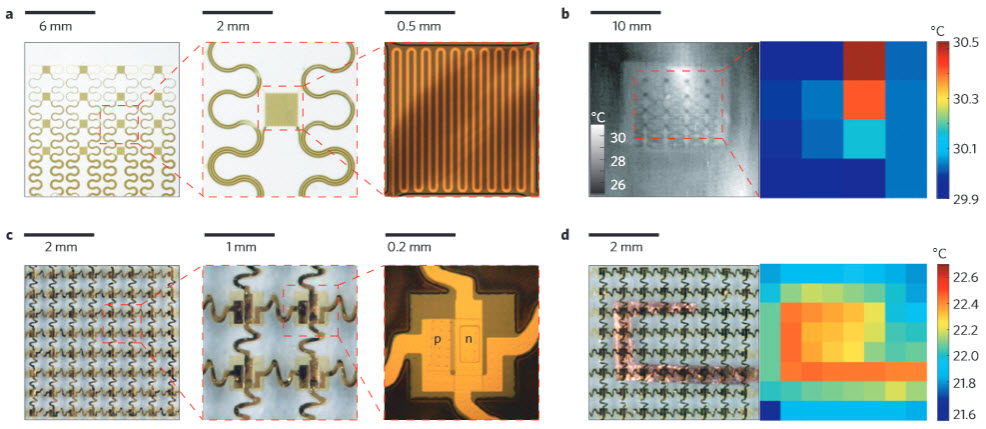
Functional demonstrations of epidermal temperature sensors and heaters. a: Optical images of a 4×4 TCR sensor array integrated on a thin elastomeric substrate with magnified views of a single sensor. b: Infrared image of a similar device mounted on the skin of the human wrist (left) and map of temperature (right), where each pixel represents the reading of one sensor in the array. c: Optical images of a 8×8 Si nanomembrane diode sensor array integrated on a thin elastomeric substrate with magnified views of a single sensor. d: Optical image of a similar device mounted on a heated Cu element (left) and measured distribution of temperature (right). (Credit: R. Chad Webb et al./Nature Materials)
In this study, the array contained heat sensors so that it could be tested for its ability to accurately detect variations in localized skin temperature when compared to the “gold standard,” the infrared camera. The profiles of temperature changes were virtually identical with the two methods.
The investigators also performed a test that is used as a cardiovascular screening procedure. Blood flow changes are detected by changes in skin temperature as blood moves through the forearm while a blood pressure cuff on the upper arm is inflated and deflated. Once again, the infrared camera and the array technology showed virtually identical temperature change profiles. Temperature was reduced when blood flow was blocked and it increased as blood was released. Slow return of blood to the forearm can indicate potential cardiovascular abnormalities.
This experiment demonstrated that the device could potentially be used as a rapid screening tool to determine whether an individual should be further tested for disorders, such as diabetes or cardiovascular disease, that cause abnormal peripheral blood flow. It could also be a signal to doctors and patients about effects of certain medications.
The final experiment addressed a feature unique to the skin array technology: delivery of a stimulus, such as heat. The researchers sent precise pulses of heat to the skin to measure skin perspiration, which indicates a person’s overall hydration. Taken together, the test results demonstrated the ability of the array technology to obtain a range of accurate, clinically useful measurements, and deliver specific stimuli, with a single, convenient, and relatively inexpensive device.
Other potential applications
In addition to heat, any type of sensors could be included, such as sensors that reveal glucose levels, blood oxygen content, blood cell counts, or levels of a circulating medication. Also, an element could be included in the circuit that delivers a medication, an essential micro-nutrient, or various stimuli to promote rapid wound healing. This ability to sense and deliver a wide range of stimuli makes the system useful for diagnostic, therapeutic and experimental purposes.
The technology has the potential to carry out such therapeutic and diagnostic functions while patients go about their daily business, with the data being delivered remotely via a cell phone to a physician – saving the expense of obtaining the same diagnostic measurements, or performing the same therapeutic stimulus, in the clinic.
Alexander Gorbach, Ph.D., one of the co-investigators from NIBIB, and head of the Infrared Imaging and Thermometry Unit, says, “We are very excited about the unique potential of this technology to vastly improve healthcare at multiple levels. Continuous monitoring outside of a hospital setting will be more convenient and cost-effective for patients. Additionally, access to data collected over extended periods, while a patient is going about a normal routine, should improve the practice of medicine by enabling physicians to adjust a treatment regimen ‘24/7’ as needed.”
The investigators are already receiving requests from other clinical research labs to use this technology, and plan to expand collaboration with academia and industry. The hope is that the research community’s interest in epidermal electronics will accelerate the development and validation of this technology and hasten its incorporation into clinical care.
Abstract of Nature Materials paper
Precision thermometry of the skin can, together with other measurements, provide clinically relevant information about cardiovascular health, cognitive state, malignancy and many other important aspects of human physiology. Here, we introduce an ultrathin, compliant skin-like sensor/actuator technology that can pliably laminate onto the epidermis to provide continuous, accurate thermal characterizations that are unavailable with other methods. Examples include non-invasive spatial mapping of skin temperature with millikelvin precision, and simultaneous quantitative assessment of tissue thermal conductivity. Such devices can also be implemented in ways that reveal the time-dynamic influence of blood flow and perfusion on these properties. Experimental and theoretical studies establish the underlying principles of operation, and define engineering guidelines for device design. Evaluation of subtle variations in skin temperature associated with mental activity, physical stimulation and vasoconstriction/dilation along with accurate determination of skin hydration through measurements of thermal conductivity represent some important operational examples.